
The FDA Is Banning Juul
Juul will no longer be permitted to be sold in the US, and the products on the market must be removed, the FDA said.

A Black Doctor Tried To Diversify Medicine. Then She Lost Her Job.
A Black doctor and her trainees risked their careers to accuse their school of systemic racism. It would be a diagnosis with no easy cure.

The White House Said My COVID Tests Would Be Fully Covered. Instead I Got Charged $43.
The United States’ messy plan for at-home COVID tests made the pandemic even more chaotic. Can we get it together before the next surge?

A Data Sleuth Challenged A Powerful COVID Scientist. Then He Came After Her.
Elisabeth Bik calls out bad science for a living. A feud with one of the world’s loudest hydroxychloroquine crusaders shows that it can carry a high price.
Un importante estudio sostiene que la ivermectina previene el COVID-19, pero los datos resultan sospechosos
Un influyente estudio argentino ha reforzado el argumento de que la ivermectina previene el COVID-19 en el 100% de los casos. Pero sus inconsistencias han llevado a expertos a cuestionar si los ensayos clínicos realmente ocurrieron tal y como se señala.
A Prominent Study Said Ivermectin Prevents COVID, But The Data Is Suspect
An influential study from Argentina has been used to argue that ivermectin prevents COVID 100% of the time — but its inconsistencies have led experts to question if it could have actually happened as advertised.

A Famous Honesty Researcher Is Retracting A Study Over Fake Data
Renowned psychologist Dan Ariely literally wrote the book on dishonesty. Now some are questioning whether the scientist himself is being dishonest.

A Cape Cod COVID Outbreak Shows The Delta Variant May Be Even More Infectious Than We Thought, The CDC Says
The Delta variant outbreak among vaccinated beachgoers in Cape Cod was “a pivotal discovery” leading to new mask recommendations, said CDC chief Rochelle Walensky.

Anthony Fauci And Deborah Birx Warned Top Officials About The “Dangers” Of Scott Atlas Last Summer, Emails Show
“I am more convinced than ever the dangers of Dr. Atlas’ views on the pandemic,” Birx wrote in an August 2020 email to Fauci and others.
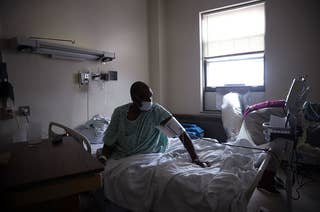
Struggling Hospitals Could Explain Why So Many Black Patients Have Died Of COVID-19
For Black patients hospitalized with the coronavirus, the quality of the hospitals they are admitted to may play an outsize role in determining whether they survive.
A Highly Controversial Alzheimer’s Treatment Won FDA Approval. Scientists Say We Don’t Know If It Actually Works.
In one sense, aducanumab is a historic drug — but many scientists say its approval is a triumph of business interests over public health.

The Editor Of A Top Medical Journal Is Leaving After The Publication Said “No Physician Is Racist”
Howard Bauchner will step down from a decade of overseeing the prestigious journal JAMA after it released a podcast questioning whether systemic racism exists in medicine.

Beto O'Rourke And Texas Democrats Are Demanding An Investigation Into The True Death Count Of The Winter Storm
No Republicans returned requests for comment.

The Texas Winter Storm And Power Outages Killed Hundreds More People Than The State Says
A BuzzFeed News analysis shows the catastrophic failure of Texas’s power grid in February killed hundreds of medically vulnerable people.

We Did It, Fam: Fully Vaccinated People Can Now Ditch Their Masks And Start Returning To Normal
Fully vaccinated people don't need to wear masks or physically distance to participate in indoor and outdoor activities, large or small, according to new CDC guidelines.
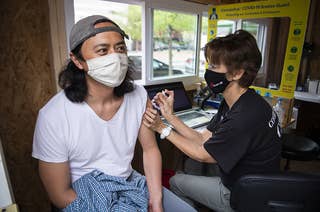
The FDA And CDC Are Restarting Johnson & Johnson’s COVID Vaccine
The vaccine, which had been paused for a week and a half, will restart after a federal advisory panel concluded that its benefits in lives saved outweigh its risks from rare but dangerous blood clots.

Johnson & Johnson’s COVID Vaccine Was Immunizing Homeless People. Then It Was Put On Pause.
Johnson & Johnson’s single-shot vaccine was in demand for some of the highest-risk people in the US. And without it, many are opting not to get a shot, clinicians say.
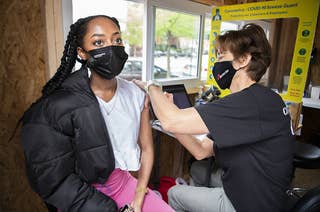
J&J’s Vaccine Is Still On Pause After A CDC Safety Panel Put Off A Vote
At least six women, including one who died, have developed an extremely rare blood-clotting syndrome after getting the vaccine.

A Top Medical Journal Said “No Physician Is Racist.” Now Scientists Are Boycotting.
After a podcast from the prestigious journal JAMA questioned whether systemic racism exists in medicine, doctors and researchers are pressing for sweeping changes.
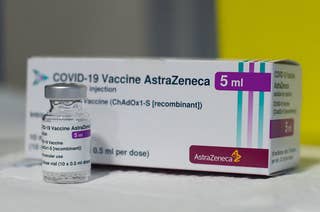
This Is What We Know So Far About AstraZeneca’s COVID-19 Vaccine And Blood Clots
So far there’s no clear evidence of a safety problem. But several European countries have paused vaccinations with AstraZeneca’s shots while investigations continue.






