In late February 2020, nine lab officials at the CDC gathered around a whiteboard to show an FDA expert how they had made the nation’s first coronavirus test — and to try to figure out why it failed.
Earlier that month, the CDC had sent thousands of faulty tests to 48 public health labs nationwide, hoping to quickly catch the first cases of COVID-19 in the US. Instead, the country succumbed to an uncontrolled spread of the new virus, without tests ready to track the outbreak. It was the opening failure of the US government's botched pandemic response, which had atrocious consequences: Hundreds of thousands of people died, millions suffered, businesses shuttered, and worthless hyperpolarized information filled the void.
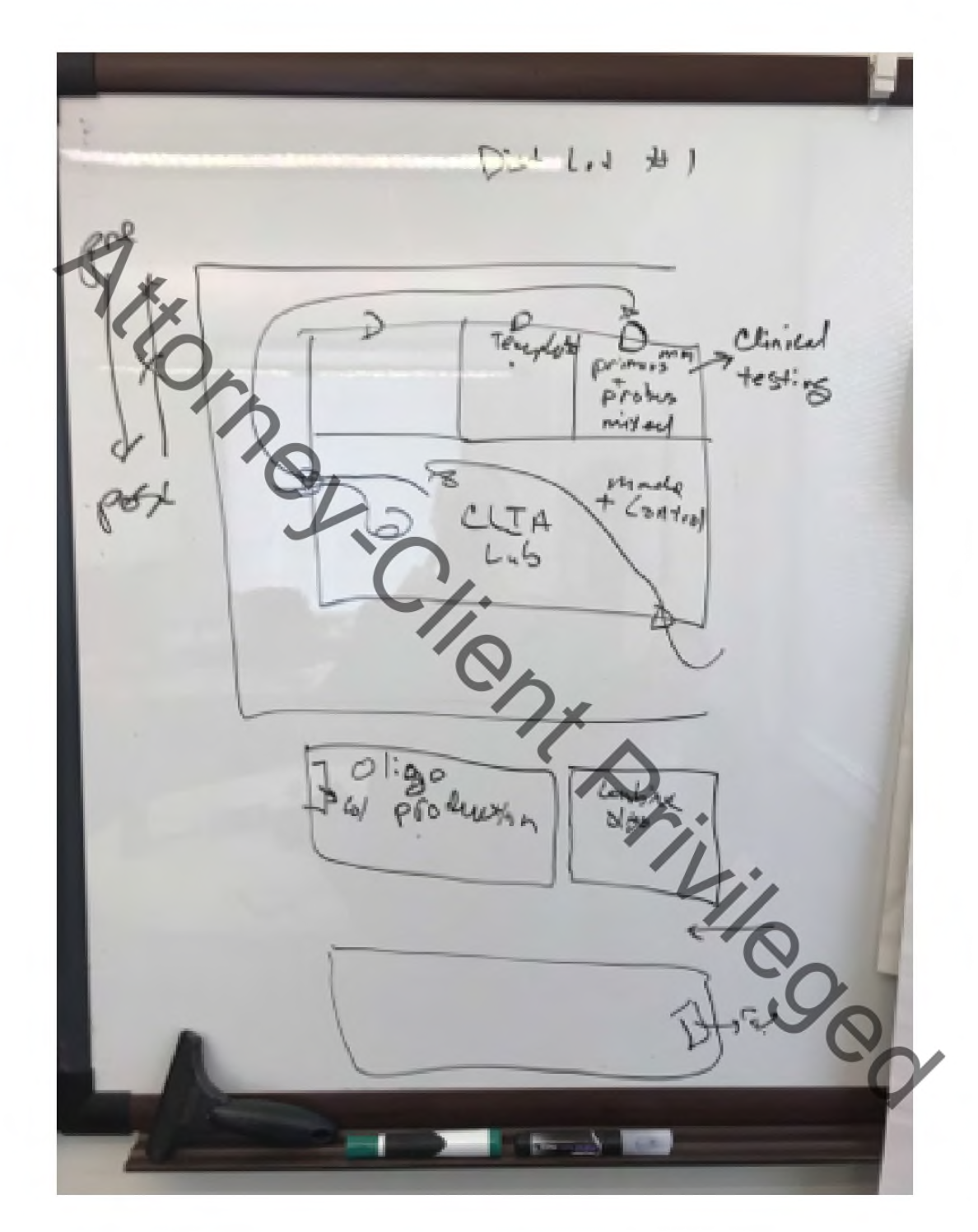
Scribbled on the whiteboard that February day was the layout of the tiny CDC lab faced with the monumental task of making the country’s first coronavirus test. Officials mapped where experiments were conducted and where people were flowing in and out. With the FDA expert looking on, some reached a damning conclusion: The lab was the perfect stage for contaminating the tests.
“Everyone had a lightbulb going off, that this is really bad,” Department of Health and Human Services lawyers wrote in an investigation completed in June 2020 and obtained by BuzzFeed News through a public records request. The cache of typed and handwritten notes, the lawyers noted, were not verbatim quotes but instead the “mental impressions” from the investigators and people they interviewed.
“People can come in doors and go,” one investigator wrote.
“No regulation of people through the lab.”
“Multiple sources [of] potential contamination.”
The US Department of Health and Human Services released a three-page summary of the investigation the same month it was completed. The full investigation documents include about 50 pages of interviews conducted by two lawyers with the HHS Office of the General Counsel, stamped “Attorney-Client Privileged,” which the health agency’s public records office sent to BuzzFeed News. An HHS official later claimed the documents were “inadvertently released” and asked BuzzFeed News to delete them, calling five minutes after an agency spokesperson turned down a request to comment on the interviews.
This investigation, based on the documents and interviews with health officials knowledgeable about the test’s development, reveals:
Government dysfunction. Congress, CDC leadership, and past presidential administrations have neglected labs at the world’s most highly regarded public health agency for decades. The small, underfunded Respiratory Virus Diagnostic (RVD) Lab was overburdened as COVID-19 blazed across the country.
Questionable blame. The initial investigation summary criticized the RVD lab for the catastrophic failure of the nation’s first COVID test. But further documents and interviews call its conclusions into question, indicating a larger lab may have been the actual culprit — something HHS investigators failed to find.
Cut corners. In the CDC’s rush to develop a lab test, officials chose to assemble two of its critical parts in the same facility, leading to cross-contamination that compromised the kits sent to state and city health agencies nationwide. This type of contamination happened before, during the 2012 MERS outbreak.
Overconfidence. The CDC did not initially rely on tests already in use in other countries, because officials thought they could build a better one. Agency leaders did not coordinate with outside manufacturers for support, and they never performed any validations of previously developed tests that could have been used as backup.
Systemic failures. The documents also reveal something even more damning: that the failures extended far beyond any one lab. The test’s problems, the documents show, are a symptom of much bigger mistakes — still unexamined — starting with the Trump administration's lack of a pandemic plan, lack of leadership and coordination at federal public health agencies, and widespread medical misjudgment in the early days of the pandemic that left the CDC holding the bag for everyone else’s failures.
The test catastrophe documented by the HHS investigation has led to unprecedented demands from former federal science officials to overhaul the CDC in order to avert another such disaster. This is a move that would essentially transform the country’s public health system. There are even calls to take pandemic testing responsibilities away from the CDC: the Biden administration proposed a $65 billion (already scaled down to roughly $10 billion in public health spending in the “Build Back Better” bill) “mission control” office to handle future pandemics. And Harold Varmus, the Nobel Prize winner and former director of the National Institutes of Health, and John Holdren, a science adviser to the Obama administration, recently called for a “COVID-19 Commission,” similar to the 9/11 Commission, to investigate the US’s blunders during the pandemic. They want to start by investigating the CDC's COVID-19 test failure.
The former director of the CDC, Tom Frieden, who reviewed the full investigation documents at BuzzFeed News’ request, said he was “kind of stunned” by the test failure. Under his tenure, in 2009, CDC rapidly developed a very successful test for the H1N1 flu pandemic, sending millions of test kits worldwide.
“The fact is CDC has phenomenal laboratories, and they’re a treasure for the US and for the world,” Frieden said, “but they clearly need to up their game.”
The investigation documents show that “now is the right time to ask broader questions about the future of the CDC,” said Scott Becker, chief executive officer of the Association of Public Health Laboratories, the group that alerted the CDC to the test’s problems last year.
Benjamin Haynes, a spokesperson for the CDC, defended the agency in a statement to BuzzFeed News: “Since the rollout of the initial COVID-19 test, CDC has implemented corrective measures and remains dedicated to the highest quality laboratory science and safety.” The US Food and Drug Administration and HHS — the FDA and CDC’s parent organization — declined to comment on the record.
As experts look to the CDC’s future, there’s much to learn from the past — especially from the early days of the pandemic, when a series of decisions at a tiny lab in Atlanta, strapped for funds and left to take on a Herculean task, would set the course for a global pandemic to hobble a global superpower.

December 30, 2019
Warnings about the coronavirus spread worldwide when the ProMED alert system, an email service to identify emerging infectious disease threats, issued an initial report that described four people in China who had developed a “pneumonia of unknown cause.”
The detection of a potentially novel virus sent health agencies worldwide scrambling to develop a test to identify infected people and stop its spread. The World Health Organization and South Korean firms quickly began designing one.
In the US, the responsibility for developing a test fell to the CDC. The agency’s headquarters, a gated and secure facility, boasted one of the four highest biosafety-level laboratories in the US and about a dozen other buildings filled with labs, technicians, and scientists. The team tasked with developing the nation’s first test was in the tiny RVD lab, which included four smaller procedure rooms, all located on the seventh floor of Building 18 at the CDC headquarters. In January 2020, the RVD lab was staffed by nine people — only three of whom were full-time employees.

By Jan. 6, 2020, one of those full-time staffers, Xiaoyan Lu, an 18-year veteran microbiologist at the lab, was already discussing the design for a test with her boss and lab chief, Stephen Lindstrom. Three days later, a virus genome released by a consortium of Chinese scientists confirmed the unknown pneumonia was caused by a coronavirus, Lu’s specialty. Neither Lu nor Lindstrom would speak to BuzzFeed News on the record about the events described in this article or about their interviews with investigators.
In the interview notes from the HHS investigation, however, Lindstrom described the development time for a CDC test as a “short fuse.” His supervisor, he said, ordered him to get FDA emergency authorization “as quickly as possible” so the test could be put to use.
So Lu began to quickly design a test from scratch. To do so, she identified three key regions of the virus genome that it could catch. Two of these regions — called N1 and N2 — were specific to the new virus. The third stretch of genes, N3, was found in all SARS-like coronaviruses. The N3 piece was chosen partly because the team had to design the test before they actually had the genome for the new coronavirus in hand, and partly as a way to keep the test effective if the virus mutated rapidly, according to Lindstrom’s notes.
On Jan. 10, the lab ordered large batches of chemicals to detect these three stretches of the virus’s genome in tested samples. These chemicals, or “reagents,” came from a much larger “core” lab at the CDC that manufactures compounds on demand for its more than 200 laboratories.
Lindstrom’s lab got batches of the three reagents — matched to N1, N2, and N3 — on Monday, Jan. 13.
But then there was a hang-up, according to Lu’s notes, with a vital fourth part of the test known as positive controls. These are copies of the part of the virus’s genes keyed to the three reagents. Every lab that gets a new test has to immediately mix the reagents with positive controls to prove that the tests can detect the virus. The copies of the virus's genes should link up with the reagents and always lead to a positive test result.
Likewise, to prove that the test doesn’t cause false positives, labs hold a separate trial of the reagents mixed with distilled water. With no virus segments in that sample, that test should always yield a negative result.
Test manufacturers usually don’t add positive controls to test kits until the final step before shipping, when everything else is freeze-dried or sealed away. That’s to avoid molecules of these viral gene bits sneaking into the reagents and contaminating them, which would cause false positives. It’s such a crucial step that some labs even manufacture positive controls at a separate site to make sure no such contamination can occur.
At the respiratory virus lab, employees felt they didn’t have this luxury, according to Lu’s notes. They needed their positive controls to check their test worked — but Lu couldn’t get them from a commercial manufacturer for another 10 days. “Can I wait a week or 10 days? No, I can’t, we need this,” the notes from Lu’s interview read. The first possible US cases were likely already spreading from China, with the Chinese New Year holiday travel in full swing.
So she asked the core lab to go ahead and make the positive controls along with the reagents. “We couldn’t get it anywhere else,” Lu’s interview notes say. And Lindstrom, according to her interview, told Lu he didn’t think that contamination would be a problem. But notes from Lindstrom’s interview disagree at this point, saying he only approved of the core lab making these early positive controls if that lab’s director were certain they could do it safely. People with knowledge of this conversation deny that Lindstrom had said he had no concerns about contamination.
Because the core lab personnel knew of the contamination risk, they made the positive controls on separate machines and in separate workspaces. Each set of these early January positive control genes were made to match the test reagents — and they contained subtle, signature differences from the genes of the “wild” virus loose in the world. They delivered them to the respiratory virus lab the afternoon after Lu’s request, at 12:51 p.m. Eastern time on Jan. 14.
“Imagine the backlash we would have had if CDC had said, ‘Hey, yeah, let’s use a German test,’” the CDC employee said.
On Jan. 16, the World Health Organization published instructions from the German Center for Infection Research for its successful COVID test. Dozens of countries worldwide began to widely manufacture and distribute it.
Why didn’t the CDC team follow suit? Lu, according to the interview notes, said that the health agency scientists thought they could make a better one. The CDC’s test mimicked the one it had made for MERS in 2012. “Our MERS assay performed better than WHO’s,” Lu recounted. And, she added, “We were racing against the clock.”
But she also knew how crucial the positive control manufacturing step was, according to her interview. CDC’s test for MERS, while ultimately successful, was initially contaminated after the lab made the same risky choice: to manufacture the reagents and controls in the same location.
A CDC employee familiar with the development of the test told BuzzFeed News that the health agency had also felt it would be better to have both US and German types of tests available worldwide — and that copying the WHO test would not have been politically possible under Donald Trump. “Imagine the backlash we would have had if CDC had said, ‘Hey, yeah, let’s use a German test,’” the CDC employee said.
January 17, 2020
The director of CDC’s National Center for Immunization and Respiratory Diseases, Nancy Messonnier, held the agency’s first public briefing on the novel coronavirus. At the time, there had been just two known deaths worldwide and fewer than 50 cases, mostly in China. The majority of the cases, she said, were thought to be due to human contact with infected animals. “Based on the information that CDC has today, we believe the current risk from this virus to the general public is low,” Messonnier said.
Nevertheless, the CDC was preparing for the worst by designing a national test for the virus. “We'll be distributing this test to state health departments,” she said.
By Jan. 18, the RVD lab had a diagnostic test for the novel coronavirus up and running.
But to manufacture and run all the tests, Lindstrom’s lab needed help. On Jan. 20, Messonnier “activated” its Emergency Operations Center at CDC to respond to the crisis and put a task force of senior officials in charge of the test kit’s manufacture. This sidelined RVD lab supervisors with coronavirus and respiratory disease expertise and replaced them with ones who had experience in flu and Zika virus outbreaks, the last large-scale global pandemics. Dozens of technicians borrowed from other CDC labs moved into the small RVD lab. For the first two weeks of developing the test, everyone in the RVD lab was in very close quarters before more space was opened up.
According to his interview with the HHS investigators, Lindstrom had “moved over to respiratory viruses to have [a] quieter life.” He had previously managed the much larger test development team at the CDC flu lab, the one that produced the H1N1 test in 2009 in record time. Now, he was trying to recapitulate that success in a smaller lab with a much smaller team.
Before January 2020, the budget for coronavirus preparedness was approximately $300,000, roughly 1/100th of the CDC budget for flu pandemic preparedness. Every year, the flu lab designs, leads, and organizes more than 1 million tests performed in public health labs nationwide. It analyzes the full genetic sequences of more than 7,000 flu samples, in support of a flu vaccination campaign; in the 2019 season, more than half the US population was inoculated.

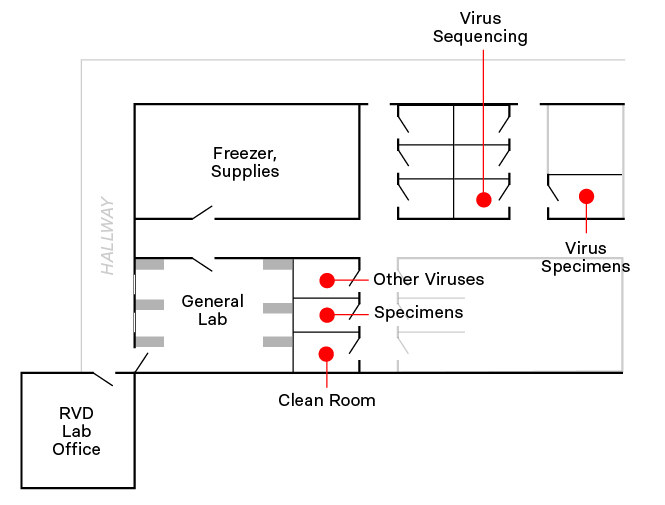
The RVD lab was underfunded despite two recent coronavirus outbreaks, SARS and MERS, that killed hundreds of people and triggered worldwide alarm. Despite decades of pandemic preparations and planning by US government officials, the CDC had kept coronavirus testing contained in this one small lab, which was also dedicated to childhood respiratory diseases and colds. One of its neighbors, the gastrointestinal diseases lab, actually had more rooms. The RVD lab initially borrowed one small procedure room from that group to handle specimens early on in the pandemic. It was later taken back.
“Steve didn’t have what he had in the flu [lab]: a system of people with knowledge, resources, staff appropriate for all stages of manufacturing qualifying design, while also doing diagnostic testing,” the investigator’s notes from Lindstrom’s interview said. Five or fewer people at the CDC were the only ones doing this for the novel coronavirus test. “Flu is very well funded, but ‘we’ are poorly funded,” Lu’s interview notes read.
On Jan. 21 Messonnier announced that the first case of someone infected with the coronavirus had been discovered in the US. He was an American who had traveled in China and had alerted his doctor about his symptoms, which developed shortly after he returned home to Washington state. His doctor notified state health officials, who shipped the man’s nasal swab overnight to the CDC. Lindstrom’s lab tested it and notified them about the positive result the next day.
“Right now, testing for this virus must take place at CDC, but in the coming weeks, we anticipate sharing these tests with domestic and international partners,” Messonnier added. By then it was clear the virus could be transmitted person-to-person.
With his cobbled-together team now running diagnostic tests, Lindstrom needed to get the FDA’s emergency use authorization (EUA) in order to ship test kits to public health labs nationwide. Getting the EUA was a big hurdle, albeit significantly slimmed down from normal approval procedures. It required the core lab to send over the reagents for 600 test kits, while the CDC team double-checked that the test worked, according to the interview notes.
By then, the city of Hong Kong was running 700 to 800 tests a day. The US had only reported 15 tests total.
People worked nights and weekends, turning out 5 to 10 times more batches of reagents than normal at the core lab, according to the investigation notes. After two weeks, Lindstrom’s lab took over making positive controls, no longer needing the core lab’s early January positive control templates to prove the test worked.
The CDC test was intended only as a tool for the public health agency to detect outbreaks, not to diagnose every sick person walking into an emergency room. Instead, in the early days of the pandemic, it became the only legal US test for every patient.
That’s in part because no one else stepped up: Large test makers, burned before by past demands for unused and unsold Ebola and Zika virus tests, had no incentive to start making tests. And smaller academic and medical center labs were daunted by the paperwork requirements to validate tests: 240 pages’ worth of patient testing data. Underlying all this hesitancy was a widely held feeling that because earlier coronavirus outbreaks had not led to pandemics, this new coronavirus would also be limited. At that time, coronaviruses were considered deadly but not terrifically infectious, as a rule.
The FDA EUA for distributing the test kits came amid the dawning realization at the end of January that the new coronavirus might spread without symptoms. By the time the CDC got the green light from the FDA on Feb. 4 to send out the test kits, China had reported more than 23,000 COVID-19 cases and over 400 deaths. By then, the city of Hong Kong was running 700 to 800 tests a day. The US had only reported 15 tests total, according to the Covid Tracking Project.

February 4, 2020
Because there was such a short supply of tests, the CDC’s guidelines in February limited testing to travelers from China with symptoms, such as a cough or fever. These criteria were cooked up in January, when the coronavirus was still considered contagious only when people had symptoms and when the virus was not known to yet be in the US — both mistakes. That meant the CDC test newly authorized by the FDA was aimed at too small a pool of people to actually spot an outbreak.
“We got the EUA 2/4,” Lindstrom said in his interview. “Shortly after released 400 kits for distribution.”
Each kit contained enough tests for 800 people. Two days later, the test kits arrived at 48 public health labs nationwide. Before they could use the tests, the labs had to use the kit materials to verify that the tests actually worked. That required testing the reagents against both the positive controls (to see if that combination reliably produced a positive result) and against distilled water (to see if they produced a negative one).
By the end of the week, Lindstrom’s lab got the numbers back. What they saw was alarming.
“N3 failed to verify in 36 of 48 labs that tried,” his interview notes said. The water test was producing false positives 75% of the time. A few labs, notably labs in New York, also reported problems with the N1 reagent.
The only tool the country had to spot and stop a COVID-19 outbreak didn’t work.

The CDC halted shipping of the tests over the weekend. The following Monday, Feb. 10, the agency held an emergency meeting among the three labs responsible for making the kits: the core lab, which manufactured the reagents, the RVD lab, which made full-gene positive controls and checked and assembled the reagents, and the Division of Scientific Resources lab, which packed everything together into test kits for shipping.
The “working hypothesis” was that the core lab had been contaminated by producing both the reagents and the early January positive controls, requiring a “deep clean,” the interview with the core lab’s branch chief, Jan Pohl, said. Pohl did not respond to BuzzFeed News’ offer to him to review the interview notes.

Meanwhile, the CDC’s screwup was becoming one of the biggest news stories in the US, sparking widespread panic over the lack of tests. By this point, South Korea was conducting 15,000 tests a day. China’s labs had started distributing test kits almost a month earlier.
At a Feb. 12 press briefing, Messonnier was peppered with questions about the troubled tests. “During a response like this, we know things may not always go as smoothly as we would like,” she said. “Of course, I hoped that this week every state — and every state did receive a kit — every state would be up and running.”
By the end of the week, only nine state or city labs could verify that the tests worked and start running them. New York state, which would soon be the site of a deadly surge in New Rochelle, was not among them.
A “deep clean” of two core lab rooms happened over the Presidents Day weekend, on Feb. 16 and 17 — a painstaking process where engineers take equipment apart and individually sterilize parts before reassembling them. ”It was important for our purification,” the interview notes read.
February 21, 2020
Days after the deep clean, FDA microbiologist Timothy Stenzel was ordered to fly to Atlanta to determine why the test was failing. He landed on Saturday afternoon and drove straight to the CDC’s headquarters — where he couldn’t get past security guards at the biosecure campus because it was a weekend.
His complaints were fielded all the way up to CDC Director Robert Redfield. Stenzel was finally let in for a tour of the RVD lab on Sunday. Over the next three days, he toured the other labs, later complaining of “vague answers” to his questions. “No one person could walk Tim through the whole process and tell him what was done,” despite the task force of senior CDC officials supposedly being in charge, Stenzel said, according to the interview notes. The notes said that at a commercial facility, versus a public health lab, “there would have been an operational lead” overseeing the process. Stenzel declined a request to speak on the record.
In those interviews, however, Stenzel reported the core lab and distribution lab appeared very clean, but the respiratory virus lab appeared “messy,” more like a research lab than a medical one — which, in fairness, is what it was. At the end of the visit, on Feb. 25, Stenzel gathered members of the three labs together to talk through what went wrong with the test. Several people pointed to the respiratory virus lab as the possible culprit, suggesting that specimens sent there early in the test’s development were contaminating the reagents.
Lindstrom was in the “hot seat” at the meeting, according to an investigator interview with CDC Division of Scientific Resources quality manager Heather Hughes. In a phone call, Hughes told BuzzFeed News she had been told not to speak to reporters by the CDC’s press office.
As Lindstrom described the respiratory lab’s workflow on a whiteboard, the “lightbulb” moment came, according to Stenzel: The RVD lab had handled virus samples and positive controls in its main room. That may have contaminated the N1 and N3 reagents handled in nearby smaller rooms as lab personnel moved around.
Normally, in order to prevent this kind of contamination, technicians handling samples need to be separated from those doing quality control on tests. Foot traffic should be one way only to prevent people from accidentally contaminating each other by contact or through the air. Under pressure and with limited space, that wasn’t happening at Lindstrom’s lab, according to Stenzel’s interview notes. Contamination under these conditions could have fouled both the N3 and N1 reagents.
Every point of this scenario — from the viral samples being opened in a common area to people tramping from contaminated areas into clean procedure rooms — is disputed by three CDC personnel familiar with the test’s development who were interviewed by BuzzFeed News. They asked not to be named, citing fear of professional retaliation by CDC.
While the core lab facilities were being cleaned and government officials were debating the source of the contamination, things were moving fast in the outside world: By the end of his visit on Feb. 25, Stenzel urged CDC leaders to move manufacturing of the coronavirus test kits out of their labs and over to a private manufacturer, according to his interview. The agency would make this move a week later.
In a CDC briefing on that same day, Messonnier jolted the nation by warning that — with 2,700 COVID-19 deaths already being reported worldwide — outbreaks would inevitably shut down work and schools in the US.
Facing what was now looking like an inescapable pandemic, the CDC’s unreliable three-part test was scrapped. On Feb. 26, the agency gave public health labs permission to nix the contaminated N3 component from tests they had already received, and it began sending out kits with only N1 and N2. The N1 reagents were no longer throwing off false positives by this point.
“We should have been permitted to do testing in January,” said Geoffrey Baird, a laboratory medicine expert at the University of Washington who reviewed the investigation notes for BuzzFeed News. “We weren’t.”
Other medical centers and academic labs across the country had also begun developing their own coronavirus tests, many looking for the N1 and N2 components like the CDC test. They had been clamoring for permission to run their own tests to speed results to doctors and meet surging demands.
On Feb. 29, the FDA finally gave them permission to use their own tests. “We should have been permitted to do testing in January,” said Geoffrey Baird, a laboratory medicine expert at the University of Washington who reviewed the investigation notes for BuzzFeed News. “We weren’t. We were waiting and had this laboratory-developed test ready, like everyone else who knew anything about this, the week after the Chinese published the sequence.”
That same day, Washington state officials reported the first suspected US death from COVID-19, a man in his 50s who had gone untested because he did not meet the CDC’s travel-based screening guidelines for the virus. They also reported the first large outbreak, diagnosed by non-CDC tests, at the Life Care Center nursing home of Kirkland, Washington. Ultimately, at least 43 residents died.
“CDC should not be in the business of manufacturing test kits,” Baird said. The RVD lab team shouldn’t have been put in that precarious position in the first place, he added. “There should be a major test manufacturing company on standby.”
The Kirkland deaths, coming just as the failure of the CDC test became clear to the public, spurred the Trump administration’s politicized response to the coronavirus.

Trump visited the CDC on March 6. Just a few hours before he’d arrived, the HHS investigators ended their three days of interviews with lab personnel. But with the cameras rolling at CDC, Trump falsely promised that “anybody that wants a test can get a test.”
In reality, only about 2,000 tests were performed on that date. Meanwhile, the country fell into uncontrolled spread of the virus.
Aftermath
Even before the investigation summary was made public, the botched tests cast a shadow over the CDC. By late spring of this year, Messonnier had retired from the health agency, and Lindstrom and other lab officials involved in the test’s rollout had moved into other positions. Messonnier, who now works at the Skoll Foundation, did not respond to a request to review and comment on the interview notes.
On June 19, a summary report was publicly released concluding that the N3 component of the CDC test was likely contaminated at the RVD lab. “Time pressure” likely played a role, the brief said. The report did not delve into deeper questions of why the lab was so small to start with, why it was counted on to run diagnostic tests for the whole country in a burgeoning pandemic, or why other tests weren’t available when it failed.
But an email that a lab official sent to CDC leadership just three days later called into question the central conclusion that the respiratory virus lab was to blame.
The email, reviewed by BuzzFeed News, stated that the report contained false statements and that HHS investigators had received genetic test results showing that the real source of the contamination was the core lab positive controls — revealed by their distinct genetic signature. In this view, the “deep cleaning” of two core lab rooms washed the contamination out of the manufacturing process. That’s why the N1 region that had failed validation at some state public health labs ran clean enough to go back into tests by the end of the month.
The investigation interviews mention that the polio lab at the CDC was running a genetic analysis of the contamination that briefly fouled up N1 reagents in March 2020. That contamination does not match the cloned coronavirus gene made by the RVD lab to serve as positive controls, BuzzFeed News learned from an internal email sent later by a CDC lab official. This is significant evidence to suggest the RVD lab wasn’t the source of the contamination — despite HHS saying it was.
HHS and the CDC did not comment on the email or the allegation that the contamination may have involved the core lab. A CDC “root cause” analysis reported last year by NPR blamed the test’s failures on the RVD lab missing quality checks on kits sent out in February. A report on the test kits, led by the Division of Scientific Resources, is slotted for a scientific journal later this month. It will suggest that the N3 reagent may have suffered from a design failure rather than from contamination, according to a lab official. That’s despite its successful use in FDA-authorized tests by major commercial companies for the rest of the pandemic.
In the aftermath of the test failure, and a critical Government Accountability Office report, the CDC has announced a series of “Lab Quality Improvement Activities,” requiring extra labs to validate tests inside and outside the agency before their release. The agency plans to ask the FDA to withdraw the initial test’s authorization at the end of the year and require all lab personnel to pass new accreditation exams by 2024.
The CDC still hasn’t officially released its own explanation for what went wrong. A separate report from the HHS inspector general started in April 2020 but has yet to be finalized. That report, which is only looking at lab procedures at the CDC and will not determine the origin of any contamination, won’t be out until next year.
The key will be making sure any changes at the CDC have lasting impacts. “A lot of money gets put in, then the emergency goes away, and people forget about it,” said Frieden, the former agency chief. “These are laboratories that need to work every day, year after year.”
The fact that the CDC was the only group in the US responsible for making tests made the failure even more consequential, Baird said. But he argued that the wider lack of tests wasn’t the CDC’s fault. Instead, he blamed the FDA, which held on to a bureaucratic EUA process that stymied fast action in the face of a public health emergency. “There was a feeling this wasn’t going to be a major outbreak early on,” a CDC lab official told BuzzFeed News, saying that senior leaders didn’t do enough to encourage industrial test manufacturers to mobilize.
An FDA official, who also spoke on the condition of anonymity, told BuzzFeed News that criticism of its EUA process is misplaced because the alternative would have been throwing the US’s doors open to a blizzard of shoddy tests in that first February of the pandemic. That would have showered doctors nationwide with false positive and negative test results in February 2020.
Overall, the test problems were just a small part of the absence of a national strategy for dealing with a pandemic, the FDA official added. A plan for nationwide testing capability, for example, was launched at the start of the pandemic and “scrapped” by the Trump administration, former FDA chief Scott Gottlieb writes in his recent book, Uncontrolled Spread: Why COVID-19 Crushed Us and How We Can Defeat the Next Pandemic. Instead, the administration had no plan aside from sending testing supplies to states and pressuring the FDA to authorize more tests after the CDC test failed.
The CDC’s test failure is a symptom of this wider pandemic failure across the entire government. There weren’t enough tests, but there also weren’t enough ventilators or personal protective equipment or swabs available nationwide.
In the aftermath, the country is left with the question of how to make sure there isn’t a similar failure in the next pandemic. Many experts are suggesting the CDC – once considered the global ideal – either needs major changes or should play a significantly smaller role.
“There is no other institution in the world that does as wide a variety of laboratory tests as CDC. And it does them under very difficult conditions, where you have huge time pressure, you have uncertainties,” Frieden said. “The fact that a problem like this could occur clearly indicates that you need additional structural reforms at CDC.”
If the Biden administration’s plan to create a central pandemic office comes to fruition, the place to locate it is probably not the CDC, “where functions are bunkered inside rigid siloes,” Gottlieb writes in his book. “Going forward, we need to provide for a national-level capability to field and deploy clinical tests for a novel pathogen.” ●
