BuzzFeed News has reporters around the world bringing you trustworthy stories about the impact of the coronavirus. To help keep this news free, become a member and sign up for our newsletter, Outbreak Today.
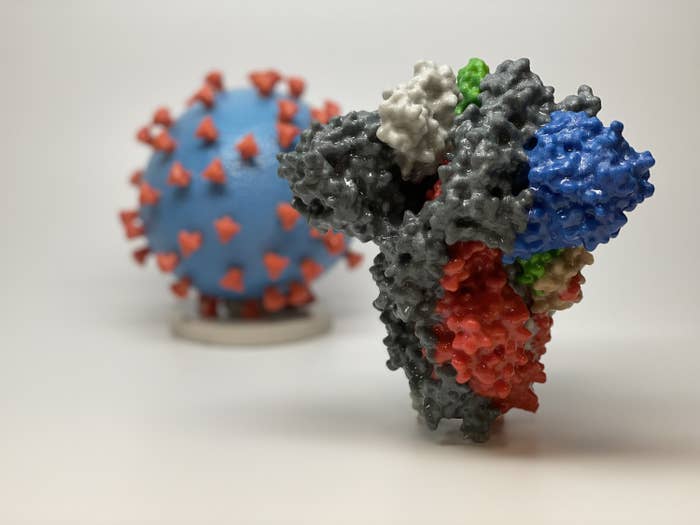
Coronaviruses take their name from the crown of minuscule spikes that bristle from their surfaces. But the new coronavirus wreaking havoc around the world has a distinctive feature on its spikes — a mysterious, loop-shaped oddity that, some researchers suggest, lets it attack a person’s lungs with deadly precision.
This small addition might also explain how the coronavirus, SARS-CoV-2, can be transmitted from person to person early on, before symptoms set in — making it so deadly it could spur a pandemic that has killed hundreds of thousands of people.
This microscopic loop has become an obsession of scientists, who are racing to decipher how the new coronavirus works and how to stop it. Figuring out how exactly the virus assaults the body is crucial for understanding what turned an otherwise common type of virus into one that can prevent people from going to work, hugging their friend, and going to the grocery store without a mask strapped across their face.
Any coronavirus that infects humans — there are four everyday types that cause 10% to 30% of colds — uses a spike as a sort of harpoon to attach to an otherwise healthy cell.
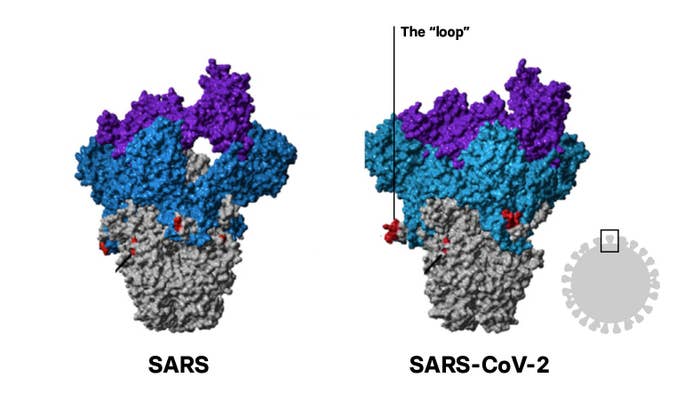
But SARS-CoV-2’s spike is a bit different. Its genes contain unique instructions that form an additional loop of protein that sits midway on the stalk, between the tip and the rest of the virus. This loop, scientists are learning, could help explain why SARS-CoV-2 is so much more dangerous to humans.
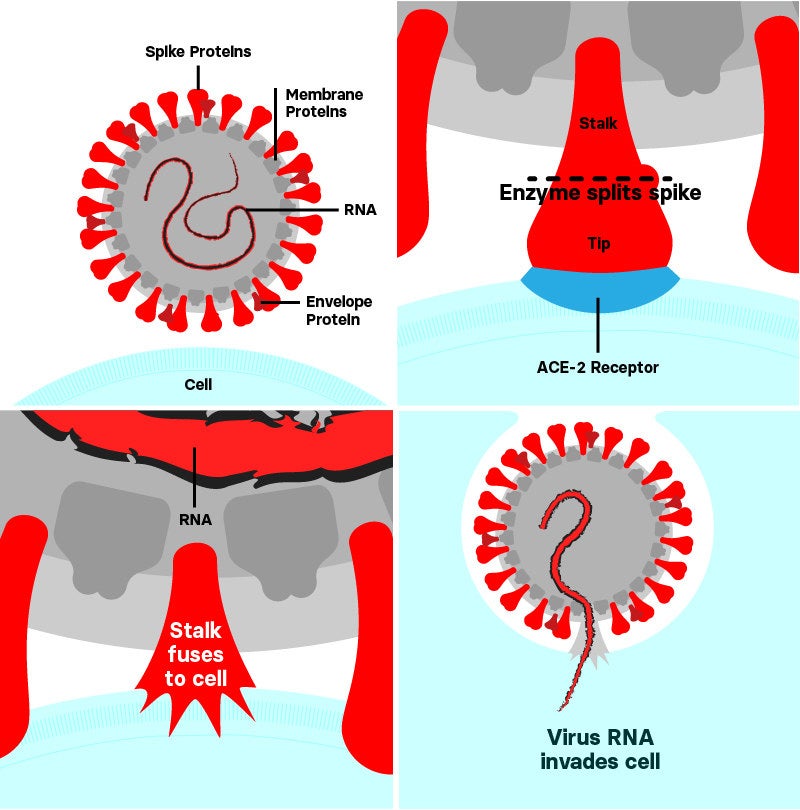
When SARS-CoV-2’s spike harpoons its prey, it docks to a receptor called ACE-2 found on the surface of some human cells.
Then, an enzyme — a substance that, in this case, sits on a human cell and causes a reaction — called furin cuts the loop, splitting the tip of the spike away from the stalk and the rest of the virus.
With the tip out of the way, the stalk then fuses to the cell and acts as a boarding ramp, pulling the virus into the cell and allowing the virus’s single-stranded genetic material, called RNA, to invade. The RNA hijacks the cell’s own machinery, turning it into a factory to churn out more viruses.
“What we’ve got is a very unique virus. It is a bit of a Jekyll and Hyde,” said Cornell University virologist Gary Whittaker, referring to the fact that it first attacks like a cold, and then turns deadly only late in an infection.
The loop of protein may help explain how differently SARS-CoV-2 infects humans compared to SARS, its closest human-infecting relative. Most cases brought on by SARS-CoV-2 are mild or symptomless, spreading infections with coughs and sneezes. But deadly cases look like SARS and its relative, MERS.
[If you're someone who is seeing the impact of the coronavirus firsthand, we’d like to hear from you. Reach out to us via one of our tip line channels.]
SARS and MERS — both coronaviruses which caused outbreaks in the past two decades — were relatively contained because infected people were usually only able to spread the virus at roughly the same time they were having severe symptoms, when the viruses were deep in the lungs. This less efficient method of attack allowed humans to know they were sick and to seek help and avoid other people before they could spread the viruses further.
With SARS-CoV-2, people become contagious early in an infection. Most people won’t know they’re sick until three to five days after exposure, while many cases present no symptoms at all, allowing for its wide and invisible spread.
SARS was more than twice as deadly as the novel coronavirus, with a fatality rate of around 11% in every confirmed case, but because it was easier to contain in hospitals, the 2003 SARS outbreak led to 774 deaths before the virus died out. In contrast, because SARS-CoV-2 spreads early in an infection, it has caused upward of 300,000 deaths worldwide.
New research suggests that the protein loop found on SARS-CoV-2 is essential to its ability to infect human lung cells. Some viruses, notably HIV, Ebola, influenza, and dengue, also rely on furin to cut their proteins down to the smaller sizes needed to infect cells. A May paper published by German researchers in the journal Molecular Cell found that mutant versions of the novel coronavirus without the furin-targeted loop were unable to infect lung cells.
“The furin site is likely needed to infect people,” Columbia University virologist Vincent Racaniello told BuzzFeed News by email. While SARS-CoV-2 most resembles a coronavirus found in bats, Racaniello noted that bat viruses don’t have a loop that’s cleaved by furin.
Drugs that block furin might help hinder a coronavirus infection, the German researchers suggested. A similar proposed COVID-19 treatment blocks a different enzyme called TMPRSS2, which chops up the spike protein even more in the very last stages as the coronavirus RNA invades the human cell.
An even more recent study of the SARS-CoV-2 spike from Whittaker and colleagues, which is still under review by the iScience journal, found that many more cellular enzymes than just furin can cut the loop and allow the coronavirus to invade cells. That adaptability — using whatever is available to start attacking cells — may partly explain why the novel coronavirus seems to infect nose and throat cells richer in these other enzymes more easily than SARS does.
Some scientists disagree that the spike protein loop is the key to the infectiousness of SARS-CoV-2. Two of the authors on the Molecular Cell study, Markus Hoffmann and Stefan Pöhlmann of the Leibniz Institute for Primate Research, said it was important to keep in mind that SARS was able to readily infect people, even without the loop. Whittaker suggested that experiments in hamsters (“a better model for the disease than mice,” he said) might provide more answers.
Meanwhile, the genetic change that brought about the loop has fueled the unfounded conspiracy theory that the coronavirus was genetically engineered in a lab.
Indian researchers first observing the furin-triggered split, in January, called its resemblance to a similar structure on HIV “uncanny,” spurring an early round of conspiracy theories that the novel coronavirus was an engineered bioweapon. That study was quickly retracted, and subsequent studies have since strongly suggested that the loop is a natural mutation, and not one cooked up in a lab and accidentally let loose on the world.
Another study, published last week in the journal Current Biology, reported a newly identified bat coronavirus, which was found in a 2019 survey of Malayan horseshoe bats. It also has a spike split similar to that of SARS-CoV-2. The new bat virus, dubbed RmYN02, is 93.3% genetically identical to SARS-CoV-2 — but its spike tip doesn’t recognize the same receptor on human cells, making it harmless to people.
This finding is “highlighting once more that the origin of SARS-CoV-2 lies in nature and not in a laboratory,” Hoffmann and Pöhlmann told BuzzFeed News, because it shows loop-like mutations can happen naturally, without any help from bioengineering.
In reality, finding a loop cut by furin in a virus “is like opening a book to a random page and finding the word ‘the’ in it — not very surprising,” University of Nebraska bioinformatics expert Kate Cooper told BuzzFeed News. She now uses the retracted paper suggesting the virus was a bioweapon, she added, “to teach my students why it is important to check your statistics.” ●
