When it comes to the biggest health story of the 21st century — COVID-19, of course — there are some glimmers of hope.
Although the Delta variant rolled through the US in late summer, pushing cases to levels not seen since early 2021 (with a corresponding rise in the death toll), daily COVID cases and hospitalizations declined in November.
But the pandemic is far from over. Given the emergence of the Omicron variant and the fact that COVID has come in waves, some experts are warning that there could be another wave coming this winter. Case numbers began rising again in early December. Deaths, more than 1,000 a day nationwide, and hospitalizations remain at severe levels among the unvaccinated.
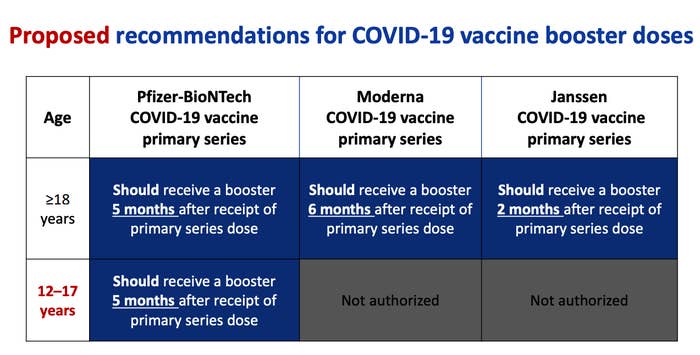
As people head indoors and vaccine immunity starts to wane — and with the emergence of potentially more transmissible variants like Omicron — cases could climb even higher. That’s led the FDA to authorize booster shots for all people 16 and older to give added protection and help prevent more outbreaks. In January, the agency added 12 to 15 year olds to that authorization for the Pfizer-BioNTech shots, a decision the CDC endorsed after an advisory panel vote, opening the door to those kids getting boosters in the US.
"We now recommend that all adolescents aged 12-17 years should receive a booster shot 5 months after their primary series," said CDC chief Rochelle Walensky, in a statement. "This booster dose will provide optimized protection against COVID-19 and the Omicron variant."
In November, the FDA first authorized, and a CDC advisory committee endorsed, a recommendation that everyone 18 and older should get a booster if they were vaccinated with a Pfizer-BioNTech or Moderna mRNA vaccine, as should all adults who received the Johnson & Johnson shot.
On Dec. 9, the FDA and CDC updated that recommendation to include 16- and 17-year-olds, who can now get a Pfizer booster six months or more after getting the second dose of the Pfizer vaccine. (The Pfizer vaccine is the only one currently recommended for children ages 5 and older.)
The Pfizer vaccine has been available to people 16 years and older for almost a year, and “its benefits have been shown to clearly outweigh potential risks,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, in a statement.
When CDC director Rochelle Walensky updated the booster recommendations in November, she cited concern about the Omicron variant to explain the decision.
"All adults are now eligible for COVID-19 booster shots," said HHS Secretary Xavier Becerra in a statement. "This is especially important ahead of the winter months, where we all spend more time indoors."
The FDA's recent decisions aim to eliminate confusion about who can receive a booster.
"I think it's a good idea, just make it easy for people to get a booster, this simplifies things," said George Rutherford, a professor of epidemiology at the University of California, San Francisco. "The concern is people who need a booster not getting one, because of confusion over their eligibility."
Following the advice of its own advisory panels, the FDA had announced in October that eligible individuals could mix and match vaccines for their booster dose. People who are 16 or 17 years old, however, are only eligible for a Pfizer vaccine for their original shots and for their booster.
“Is there going to be a wave this winter? There will undoubtedly be a wave this winter among people who are unvaccinated,” Rutherford said. “But at least taking care of this problem of waning immunity will blunt the number of cases among people who have been fully vaccinated. So I think that’s good. There will be less morbidity and mortality because of that.”
However, if you are unvaccinated, “you are hanging out in the wind,” Rutherford said. “Again, my only advice is to get vaccinated.” (Check out this vaccine tracker to see how many people have been vaccinated.)
Here’s what you need to know about what booster shots are, when you should get them, who is eligible, whether you can mix and match vaccines, and the side effects you might experience.
Boosters work.
In general, if you have a healthy immune system, COVID vaccines can be more than 90% effective when it comes to preventing symptomatic COVID, hospitalizations, and death. (The one-shot J&J vaccine has typically been slightly less effective than the two-shot Moderna and Pfizer vaccines.)
But protection can wane over time. A July 2021 study in Nature Medicine estimated that COVID vaccines with a 90% efficacy dropped to 70% in about seven months and 50% at about nine months. The drop in protection is caused by antibody levels decreasing over time, but also because of the highly contagious Delta variant, now the dominant strain in the US. But many scientists have argued that the vaccines are still highly protective for people who are not at high risk.
In September, the FDA granted Pfizer permission to offer boosters for those at higher risk based on evidence that protection started to wane at four months. A study of the Pfizer vaccine found that protection against all variants and the Delta variant was 97% and 93%, respectively, one month after vaccination, but it declined to 67% and 53% four months later. Moderna also published research suggesting that protection wanes over time. People who got the Moderna vaccine during the previous eight months had 36% fewer infections than people vaccinated a year ago.
A booster shot can help. FDA data suggests that getting one can reestablish strong protection against the virus. Pfizer, for example, claimed that a booster dose restored the vaccine to about 95% efficacy within a week, which is consistent with observations from Israel.
“The available data suggest waning immunity in some populations who are fully vaccinated,” acting FDA Commissioner Janet Woodcock said in an announcement on Oct. 20. “The availability of these authorized boosters is important for continued protection against COVID-19 disease.”
The FDA and CDC have approved boosters for people 16 and older.
The FDA has authorized booster doses of the Pfizer or Moderna vaccines for anyone 18 and older who finished their initial two-dose vaccination series at least six months ago, or received the one-dose Johnson & Johnson vaccine at least two months ago.
People who are 16 or 17 who completed their vaccination at least six months ago are also eligible for a booster shot, but can only receive it with the Pfizer vaccine.
You should get a booster if you are 65 or older.
In general, whether you’ve had the Pfizer-BioNTech, Moderna, or J&J vaccine, you should get a booster if you are 65 or older. This is regardless of your job, living circumstances, or underlying health conditions.
The risk of getting seriously ill or dying from COVID-19 goes up as you age, starting in your 50s and increasing in the 60s, 70s, and 80s. People who are 85 or older are at particularly high risk.
You should get a booster if you have a health condition that puts you at high risk.
If you are an adult who’s younger than 65 but you have an underlying health condition, you should get a booster — particularly if you are 50 or older.
That includes people who are pregnant, obese or overweight, or immunocompromised and those who have a health condition such as:
- Cancer
- Chronic kidney, liver, or lung diseases (such as moderate or severe asthma)
- Dementia
- Diabetes
- Down syndrome
- Genetic conditions, such as sickle cell disease or thalassemia
- Heart diseases
- High blood pressure
- HIV
- Mental health conditions such as depression or schizophrenia
- Stroke
- Tuberculosis
People who smoke, people who used to smoke, and those with substance use disorders, like alcoholism or opioid use disorder, should also get a booster. (The CDC notes that this list isn’t the final word; there are other conditions that put you at risk, so talk with your doctor about your own personal risk.)
The risk is higher for people with health conditions who are 50 or older, so they are strongly encouraged to get boosters.
If you are at risk due to your job.
Other people who might most need a booster are those who work in occupations that put them at risk for COVID-19.
That includes:
- Healthcare workers
- Firefighters
- Police
- Educators, such as teachers, daycare workers, and support staff
- US postal workers
- Public transit workers
- Food and agriculture workers
- Those who work in grocery stores, corrections, or manufacturing
If you are at risk due to where you live.
People who live in high-risk congregate settings like homeless shelters, prisons, jails, and healthcare or mental health institutions are at higher risk of COVID and might more heavily consider getting a booster.
If you are pregnant, you can get a booster.
People who are pregnant, recently gave birth, or who are thinking about getting pregnant should get vaccinated. They are also eligible for booster shots.
As of the end of Oct. 11, more than 22,000 people who were pregnant have been hospitalized and 180 have died due to COVID, according to the CDC. People who are pregnant and have symptomatic COVID are twice as likely to be hospitalized and have a 70% higher risk of dying than people who are not pregnant, the federal agency said.
“COVID-19 vaccination is recommended for people who are pregnant, breastfeeding, trying to get pregnant now, or might become pregnant in the future,” according to the CDC.
The partners of people who are trying to get pregnant should also get a vaccine.
All adults who got the Johnson & Johnson vaccine can get a booster.
The FDA advisory panel has unanimously recommended that anyone who is 18 or older and received a J&J vaccine get a booster two months after they received their dose.
The J&J vaccine has advantages in that it’s one shot instead of two. But although it’s highly effective at protecting against COVID, it hasn’t been quite as effective as the other vaccines available in the US.
For example, CDC data suggests that when it came to preventing COVID hospitalizations, Moderna was 93% effective, Pfizer was 88% effective, and the J&J vaccine was 71% effective.
Some FDA advisers even suggested that the J&J vaccine should have been a two-shot vaccine from the start.
“I think this frankly was always a two-dose vaccine," said FDA vaccine advisory panel member Paul Offit of the Children’s Hospital of Philadelphia. "It’s hard to recommend this as a one-dose vaccine.”
“It’s always been treated like the poor cousin of the mRNA vaccines and I think that’s unfair,” said Dr. Peter Hotez, codirector of the Center for Vaccine Development at Texas Children’s Hospital and dean of the National School of Tropical Medicine at Baylor College of Medicine. “I think the benefits of the mRNA vaccine are probably a bit inflated and the disadvantages of the J&J vaccine have been a bit inflated.”
Adults can mix and match boosters.
If you got a J&J vaccine, can you get a Pfizer or Moderna booster?
Yes, if you are 18 or older. Research suggests that might be a good approach: A National Institutes of Health study presented in October to the FDA advisory committee found that a Moderna booster given to people who had previously received a J&J shot produced the most antibodies against the coronavirus. A Pfizer booster given to people who had received a J&J shot produced slightly fewer antibodies. A J&J booster produced the least antibodies.
That surprised the FDA’s advisers, who noted that standard practice is to boost a vaccine with its original shot to amplify the immune reaction it first triggered.
However, Hotez noted that the study ended at 30 days, which “tends to stack the deck against seeing the benefits of the J&J vaccine,” he said.
The J&J vaccine “gives you kind of a delayed kinetics so the antibody titers rise later,” Hotez said. More research may show J&J is more effective than seen in the study.
The FDA’s authorization in October also allowed eligible individuals who received Pfizer’s or Moderna’s vaccines to mix and match booster shots. People who received two doses of a Pfizer or Moderna vaccine can get any of the three authorized vaccines as their booster dose.
There’s no real guidance on what booster shot you should get. The best combination of vaccines is still unclear, and the federal agency’s recommendations reflect that.
“We wanted to provide a lot of flexibility because some people don't know what vaccine they got in certain circumstances,” Woodcock said at a press conference in October. “There are people who don't know, there are people who do not want to get the same vaccine they got before because they had some sort of reaction.”
Any one of these combinations is reasonable, Marks said.
It’s very important that anyone who is in an eligible group get a booster, Woodcock said. “We would expect that many folks will continue with the same regimen that they started with.”
When you go to get a booster, you may be asked some questions to determine your eligibility, but you won’t necessarily need to provide medical verification. “They’re taking people at their word that they fall into one of the categories,” Marks said.
When you should get a booster depends on the type of vaccine you received.
If you are eligible to get a booster, you can get it at least six months after a Moderna or Pfizer vaccine. As mentioned, the CDC advisory panel recommended that adults 49 and younger "may" get a booster, not that they "should," in this group.
Anyone who received a J&J vaccine should wait at least two months before getting a booster.
Boosters are a bit different from the “regular” vaccine.
A booster isn’t necessarily just one more dose of a “regular” vaccine. It may be the same dose or it may be lower.
If you received a Pfizer vaccine, you got two 30-microgram doses at least 21 days apart. The booster is also 30 micrograms.
If you received the Moderna vaccine, you got two 100-microgram doses at least 28 days apart. The booster is a 50-microgram dose, or a half dose of one of your original vaccine shots. Moderna found that that dosage is sufficient, triggering a fourfold rise in the protective response against the Delta variant, according to one study.
Moderna is also looking at future shots designed more directly against the Delta variant since it now composes 99% of US cases and looks likely to be the basis for future variants.
The J&J booster is the same as the first dose, and it’s given at least two months after the shot.
Some people need a third shot, not a booster.
What’s the difference, you ask? A third shot is just that — the exact same dose as the first one or two, often given sooner than a booster — and it’s given to people who don’t have a robust enough response the first time around.
Since August, the CDC has strongly encouraged people who have moderate to severe immune system problems to get a third shot.
That includes people who have had an organ transplant, are currently getting cancer treatment, have HIV, or have to take immune-suppressing medication for autoimmune disorders and other conditions.
Of people hospitalized for COVID in June and July, 86% were unvaccinated and 14% were vaccinated, according to CDC data. People with weaker immune systems make up 40% to 44% of the fully vaccinated people who need to be hospitalized.
The death of Colin Powell, the first Black secretary of state for the United States, highlights the COVID risk for people with compromised immune systems. Powell, 84, was fully vaccinated but also had multiple myeloma, a type of cancer that can severely affect the immune system. In addition, the treatments for multiple myeloma, and other types of cancer, can reduce the efficacy of vaccines.
If you have a weakened immune system, your third shot should be at least 28 days after the second dose of a Pfizer-BioNTech or Moderna vaccine. (In other words, you don’t have to wait six months as you do for a booster.)
There is not enough data at this time to know whether a second dose of a J&J shot protects immunocompromised people in the same way a third dose of a Pfizer or Moderna shot does, according to the CDC.
Booster side effects are similar to getting a second vaccine shot.
You may have experienced side effects after being vaccinated, particularly after getting the second dose of the Pfizer or Moderna vaccine. Symptoms like a headache, fever, or nausea can be a sign your immune system is reacting to the vaccine and churning out the protective antibodies you need.
When it comes to the booster shot, studies have indicated you might have symptoms similar to what you experienced with the second dose of a Pfizer or Moderna vaccine.
According to the FDA, most side effects were mild or moderate and included headache, fatigue, and muscle aches.
More people said they had swollen glands and tenderness in the armpits after the booster than after the first vaccine.
“Same as your second dose — sore arm and feeling flu-ish, but that’s about it,” Rutherford said. “You might want to take the next day off.”
In presentations to the FDA’s vaccine advisers, Johnson & Johnson reported side effects that were mild and similar to or lower than those seen after the first shot. However, the data was very limited — measured in fewer than 40 people — and the FDA has not had time to double-check the company’s data, something that the agency said it would follow up on.
The most common side effects reported after a Johnson & Johnson booster shot were headaches and tiredness.
It takes a couple of weeks for booster protection to kick in.
Your body needs a little time to make the antibodies you need to fight off COVID. So a booster isn’t instant protection.
The first round of vaccines can take a couple of weeks to kick in, and a booster shot is the same.
However, longer-lived “memory” immune cells are still in your body from the initial vaccination. For the vast majority of people, these can help protect against getting very sick or being hospitalized before a booster starts to help.
In most people, immunity to the virus that causes COVID will start to rise in the weeks after you’ve had a booster shot. The Pfizer and Moderna booster shot effects reached their most effective point after 15 days, while the Johnson & Johnson booster increased steadily for at least 29 days afterward, and possibly longer.
COVID boosters are free.
Yes, they say nothing in life is free. But boosters for COVID vaccines are, indeed, at no cost. And they are widely available.
“These boosters are free,” President Joe Biden said at a press briefing in October. “I’ll say it again: They’re free, available, and convenient to get.”
You are still considered fully vaccinated even if you don’t get a booster.
If it’s been two weeks since your second shot of a Pfizer or Moderna vaccine or two weeks after your single shot of the J&J vaccine, you are considered fully vaccinated.
After that, even if you don’t get a booster, you are still fully vaccinated, according to the CDC.
And finally, if you haven’t been vaccinated at all, get the shot — even if you have had COVID already.
The pandemic is still ongoing, so it’s a good idea to get vaccinated if you haven’t already.
“I think we’re in the eye of the hurricane again,” Hotez said. “We’ve seen the exact same pattern last year where it started going down around this time, and then as we moved towards the Thanksgiving holidays, then it started going back up.”
If you have recovered from a COVID infection, you have some immunity to the virus.
However, it may not be as much protection as the vaccine provides: A study in Kentucky suggested that unvaccinated people who weathered a past COVID-19 infection were 2.34 times more likely to get infected again than vaccinated ones, although research is ongoing.
“I think there are still too many unvaccinated people, so the recommendation is to go ahead and get vaccinated,” Hotez said.
All of the vaccines are good choices, he said. “I think they are all pretty equivalent in terms of safety profile and efficacy.”
This is a developing story. Check back for updates and follow BuzzFeed News on Twitter.

