The journalists at BuzzFeed News are proud to bring you trustworthy and relevant reporting about the coronavirus. To help keep this news free, become a member and sign up for our newsletter, Outbreak Today.
A Stanford University professor’s wife invited parents in a wealthy enclave of Northern California to sign up for her husband’s coronavirus antibody study this month, falsely claiming that an “FDA approved” test would tell them if they had immunity and could “return to work without fear,” according to an email obtained by BuzzFeed News.
The email, sent to a listserv for Ardis G. Egan Junior High School in the city of Los Altos on Thursday, April 2, advertised a study set to begin the next day. With the subject line “COVID-19 antibody testing - FREE,” the email described how participants could gain “peace of mind” and “know if you are immune.” The results would help researchers calculate the virus’s spread throughout the surrounding county of Santa Clara, according to the message sent by Catherine Su, a radiation oncologist married to Jay Bhattacharya, the Stanford professor of medicine leading the study.
Weeks later, early results from Bhattacharya’s team would conclude that, based on the tests, the area had 50 to 85 times more infections than reported cases. That finding, along with their claim that the coronavirus would therefore have a lower fatality rate than previously thought, made national headlines. But almost as immediately, the study came under fire from scientists, who said it was based on a heavily flawed data analysis that ignored questions about the antibody test’s false positive rate — as well as a problematic Facebook recruitment strategy.
The email reveals that the researchers did not disclose another way participants were recruited that could have further skewed the results. In addition to targeting a specific demographic of parents in a wealthy part of Silicon Valley — making it even less likely that the participants represented a random sample — the email falsely claimed that the study’s antibody test was FDA approved, and was worded in a way that might have disproportionately attracted people who had previously been sick. It also misrepresented what participants could learn about their health from the testing.
Given how new this coronavirus is, scientists do not definitively know how long antibodies can protect you from getting sick again. Yet Su’s email stated: “If you have antibodies against the virus, you are FREE from the danger of a) getting sick or b) spreading the virus. In China and U.K. they are asking for proof of immunity before returning to work. If you know any small business owners or employees that have been laid off, let them know -- they no longer need to quarantine and can return to work without fear.”
Marm Kilpatrick, an infectious disease researcher at the University of California at Santa Cruz, told BuzzFeed News these statements were “super concerning.”
“They're offering people information about their possible immunity that I don’t think they can accurately give them, that might put these people in danger if they take actions like interact with people without social distancing in unsafe ways,” he said.
Asked for comment, Bhattacharya said that his wife’s email was not associated with the research team’s work. “The email you reference was sent out without my permission or my knowledge or the permission of the research team,” he wrote in an email to BuzzFeed News. He said that he believes the note was also shared on social media sites.
Bhattacharya acknowledged that the email skewed the makeup of the study’s participants, but argued that the researchers corrected for the difference in volunteers. “Our tracking of signups very strongly suggests that this email attracted many people from the wealthier and healthier parts of Santa Clara County to seek to volunteer for the study,” he wrote. “In real time, we took immediate steps to slow the recruitment from these areas and open up recruitment from all around Santa Clara County.”
He added that a revised preprint would be released in the next two days and would address criticisms of the study’s sample selection.
Su did not return requests for comment.
On Monday, Stanford spokesperson Julie Greicius said in a statement, “Stanford Medicine is aware of news reports regarding the recruitment of participants for the recent COVID-19 seroprevalence study in Santa Clara County. These matters are being handled by the appropriate committees at Stanford.”
Since last week, when the Stanford team posted a non-peer-reviewed preprint online, they’ve stirred debate with their findings: that as of early April, in contrast to the 956 cases reported in Santa Clara County, there were an estimated 48,000 to 81,000 infections that had gone undetected. And while the death rate from confirmed cases in the US is over 5%, due in part to a lack of diagnostic testing, the researchers said their results suggested the true rate, from diagnosed and undiagnosed cases, was drastically lower: between 0.12% to 0.2%.
Their study, based on tests administered to 3,330 people, is one of the biggest antibody surveys in the US so far. Scientists around the world, from New York to Germany to Italy, are counting on such tests to help answer one of the most important questions about the virus: how widespread, exactly, it is. Results from these early surveys vary widely.
Antibody tests, also known as serology tests, differ from the nasal- and throat-swab tests that can spot a current infection. Instead, these look for signs in the blood that the immune system had fought off an infection in the past. Government officials are touting them as key to reopening the economy and helping identify who is safe from reinfection and safe to go back to work.
But there are limits to what antibodies can reveal. With other infectious diseases, antibodies signal a measure of protection against getting sick again. No one knows if that is true of this virus, or, if it is, how long such immunity might last.
There are also potential problems with the antibody tests themselves. To quickly increase availability, the FDA is allowing tests to be sold without the agency independently validating their accuracy claims. In the US, only four tests have “emergency use authorization” indicating the agency has reviewed their data — and more than 130 others have unvalidated accuracy rates.
One of these unauthorized tests was used for the Santa Clara County study, as well as for a recent, similar study in Los Angeles County.
But Su’s email to parents at Egan Junior High, which is attended by more than 650 seventh- and eighth-graders, falsely described the test as an “FDA approved antibody test for Coronavirus.”
In fact, the test was distributed by Premier Biotech in Minnesota, and made by Hangzhou Biotest Biotech of China. As NBC News has reported, China recently banned the test, along with other COVID-19 tests, from being exported because it hadn’t been vetted by China’s equivalent of the FDA. (The tests used in the two studies were shipped to the US legally, before the ban, according to Wired.)
Bhattacharya did not respond to questions addressing the false claims made about the test.
Then there’s the question of how to accurately calculate a virus’ prevalence in a community. To do so, it helps to have underlying test samples from people who are representative of their community as a whole.
If, for instance, tests are disproportionately done on people who were sick in the past, their positive samples can skew the prevalence estimates higher than the true number.
The April 2 email asked for “HEALTHY volunteer[s].” But it also mentioned that “the disease likely entered the California in December (based on 2019 travel data),” due to travel between the US and Wuhan, China, at that time. And the email explained that “the serum antibody test determines whether your immune system has fought off the virus and created antibodies to protect you from future exposure.”
Those statements might make the study sound especially appealing to people who had COVID-19 symptoms in the last few months, but weren’t able to get tested.
“It’s troubling in that it seems the wording might bias people to be more likely to participate in the study if they thought they have antibodies against the virus,” Kilpatrick said.
Bhattacharya said that the issue of sample selection bias would be addressed in the forthcoming revision.
In their preprint, the researchers said that they’d recruited participants through locally targeted Facebook ads, which led people to a survey that “provided information” about the study. They didn’t share the ads’ language.
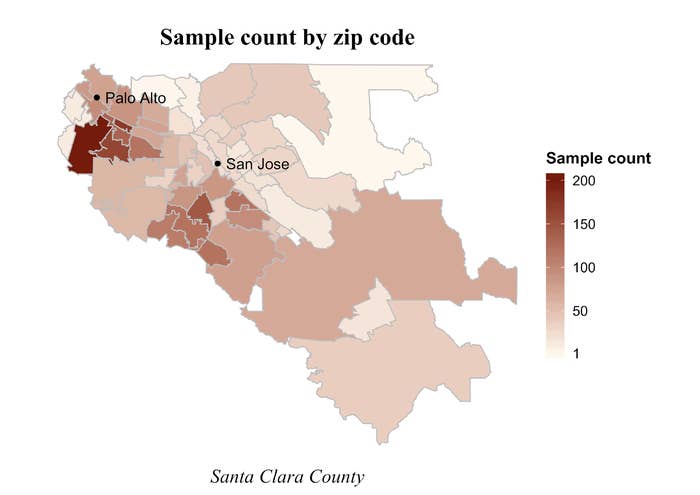
The study ended up recruiting a group that wasn’t representative of the county as a whole in some ways. Some zip codes were more represented than others — with zip codes in Los Altos making up some of the highest concentrations of participants, according to a figure in the preprint. Bhattacharya acknowledged that the email had contributed to this effect, but argued that he and his team had corrected for the imbalance in their statistical analysis by adjusting for the difference.
At the bottom of the email, Su explained that participating would “contribute to knowledge of the prevalence of virus spread in Santa Clara County.”
For further reading, she linked to a March 24 Wall Street Journal op-ed that argued the number of true infections from the coronavirus was likely much higher than reported, and its fatality rate was therefore likely to be much lower. The authors were two Stanford professors, one of them her husband.
CORRECTION
The email was sent on Thursday, April 2. An earlier version of this story misstated the day of the week it was sent.
UPDATE
This story has been updated to include a comment from Stanford University.
