BuzzFeed News has reporters across five continents bringing you trustworthy stories about the impact of the coronavirus. To help keep this news free, become a member and sign up for our newsletter, Outbreak Today.
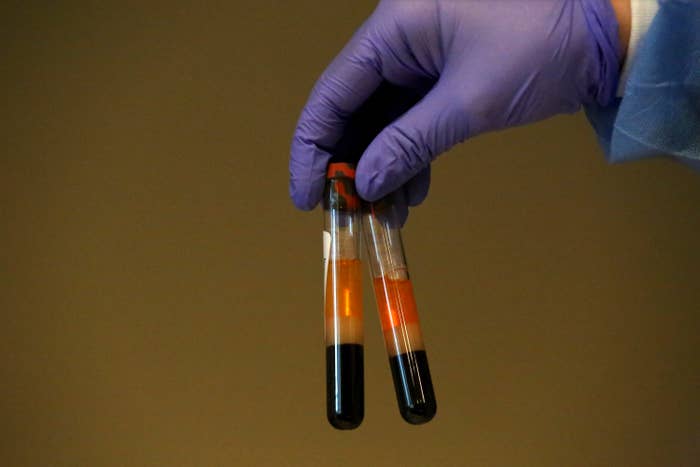
In a reversal of a policy that led to dozens of unauthorized coronavirus antibody tests flooding the US market, the Food and Drug Administration said Monday that manufacturers must submit data proving the accuracy of their tests within the next 10 days or face being pulled from the market.
In mid-March, the agency had loosened its regulations of antibody tests, which scan people’s blood for signs of a past infection, in an effort to quickly increase test availability. The move allowed test manufacturers to bypass the process for “emergency use authorization,” which requires that the agency review data about the tests’ accuracy.
Instead, companies only had to tell the FDA that their tests were for sale and that they had internally validated them. As a result, there are now 12 authorized tests — and more than 165 that are not, according to the FDA’s website.
The move led to what many commentators have described as a “wild west” in antibody testing. Some tests have been marketed with misleading claims, and others have provided inaccurate results.
In two high-profile instances, large numbers of tests purchased from Chinese manufacturers by the UK and Indian governments were later rejected for providing inaccurate or unreliable results. But those tests — reportedly manufactured by Guangzhou Wondfo Biotech, Zhuhai Livzon Diagnostics, and Hangzhou AllTest Biotech — are still available for purchase in the US, according to the FDA’s website.
“We unfortunately see unscrupulous actors marketing fraudulent test kits and using the pandemic as an opportunity to take advantage of Americans’ anxiety,” the FDA said in a statement.
The policy had been widely criticized by public health experts who were concerned that the tests would give the public false information about their exposure to the virus at a critical juncture when government officials are debating how to safely restart the economy and some states are already beginning to open up.
“We’ve long been concerned that allowing tests on the market that have not been approved and authorized for use is a recipe for disaster,” said Scott Becker, CEO of the Association of Public Health Laboratories, in a statement. “This revised policy makes a lot of sense and should have been in place over the last six weeks.”
For the public, the allure of antibody tests — that a positive result would mean protection from reinfection — has been largely outweighed by questions about what information the tests can provide. To begin with, scientists do not know whether (or for how long) having antibodies confers immunity.
On top of that, health care providers have struggled to figure out which tests are reliable, and patients are unsure if they can trust their results. An unverified test was used in a pair of controversial studies that sought to measure the virus’s spread in communities in Northern and Southern California.
Experts are mainly concerned about the possibility of false positives, which would erroneously indicate to people that they’d recovered from the virus. That might give them the unfounded confidence to stop socially distancing and potentially contract the disease or spread it to others. A team of scientists recently evaluated a dozen antibody tests and found that only one never delivered a false-positive result.
“The worst thing you can do with a test is have a test that gives false positives,” Paul Bates, a virologist at the University of Pennsylvania, told BuzzFeed News. “I think that’s a shortcoming of the tests that are being pushed out there right now — they haven’t been thoroughly vetted.”
Now, companies have 10 business days to submit their validation data to the FDA, and the agency has recommended specific accuracy benchmarks for developers to meet. The agency said that almost half of the tests offered by manufacturers are already undergoing review for an emergency use authorization or are in the early stages of seeking an authorization.
“I applaud the decision,” said Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota by email. “We need detailed data on test performance characteristics before [they are] approved.”
