
Amid a global tragedy, the striking effectiveness of vaccines against the coronavirus stands out as one of the pandemic’s few good-news stories for humanity.
And the vaccines are the success story that, so far, has kept on going. Vaccinations are proving to be just as effective in the real world as they were in clinical trials, while remaining highly protective against the more contagious variants spreading worldwide. And two recent studies found that immunity to the virus could last for years.
While some viruses, most notably HIV, have eluded vaccination efforts for decades, SARS-CoV-2 has turned out to be an ideal target.
“It’s going to sound odd, but the truth is, we humans sort of lucked out,” said James Musser, a pathologist at Houston Methodist Hospital. “It doesn’t always go this way with vaccines.”
In November, initial reports of the stunning 95% efficacy of the messenger RNA (mRNA) vaccines produced by Pfizer-BioNTech and Moderna came as a breakthrough moment just ahead of a crushing surge in US cases. Those results were far above the 50% efficacy threshold for shots set by the FDA in October of last year, and they well exceeded the 70% to 75% bar that some, including Anthony Fauci, chief of the National Institute of Allergy and Infectious Diseases, had initially hoped for.
“Nobody really expected it to be this good,” said vaccinologist George Rutherford of the University of California, San Francisco. “It’s remarkable. It’s why we invested in molecular biology for the last 40 years.”
But getting everyone immunized with these remarkable vaccines remains a huge challenge. Even as the US returns to more normal life, with more than 40% of the population fully vaccinated and a goal of 70% having at least one shot by July 4, the rest of the world continues to face severe outbreaks. Less than 6% of the global population has been vaccinated, with more than 60 countries having administered fewer than five shots for every 100 people in their population. Globally, at least 3.7 million people have died of COVID-19.
That's left countries clamoring for more vaccine options, even ones that are less effective than those authorized in the US. On Monday, the World Health Organization authorized China’s CoronaVac shots, an older kind of vaccine made from inactivated viral particles, for its global vaccine-sharing program. It is only about 50% effective in preventing an infection, but studies show it reduces the risk of severe disease.
“The world desperately needs multiple COVID-19 vaccines to address the huge access inequity across the globe,” the WHO’s Mariângela Simão said in a statement announcing the authorization. And on Thursday, the Biden administration unveiled a plan to share 25 million vaccines worldwide, three-quarters of them through the WHO’s COVAX program, the first step toward sharing at least 80 million doses by the end of June.
“The basic bottom line on this is that the United States is not doing this as some kind of back-and-forth deal where we're getting something for what we're giving,” national security adviser Jake Sullivan said at the White House briefing announcing the change. “We are giving these for a single purpose; it is the purpose of ending this pandemic.”
That we can talk about ending the pandemic with vaccines at all is partly due to good luck and partly due to decades of research preparing for such a deadly virus.

A Perfectly Normal Virus
Vaccines have been a triumph of science in the last century, eradicating smallpox and drastically curbing polio, rabies, and measles. But there have also been disappointments: We still have no vaccines against HIV or malaria despite decades of research. So in early 2020, scientists were unsure whether SARS-CoV-2 would be a virus we could fight with a vaccine.
“There was a lot of fear early on that this virus was doing something very strange,” said Shane Crotty, a virologist at the La Jolla Institute for Immunology. The novel coronavirus attacked in a confusing way, causing no symptoms in some people and proving deadly for others. This suggested a complex interplay with the human immune system that would make it hard to design a vaccine against.
A little over a year ago, however, Crotty’s lab released data from recovered COVID-19 patients suggesting that their immune responses were similar to those seen after infections with other viruses. People who recovered first made antibodies to the virus and then generated longer lasting immune cells called T cells and B cells that retained a memory of the virus to fight off future infections. Vaccines spur the same response, only without causing any illness. This was an early sign that vaccines could be effective against the new virus.
“We could say, ‘This looks like a perfectly normal virus, we are playing by rules we understand,’” Crotty said. But the immune system is labyrinthine, and often surprising, in its complexity. So it was only when early safety studies of vaccine candidates were released last summer — showing a level of antibodies postvaccination that was roughly eight times higher than those seen after a natural infection — that things started to look really encouraging.
And scientists knew that, unlike the flu, which sloppily shuffles its genes when it reproduces, the coronavirus is a more sophisticated bug, repairing errors that crop up in its genes during replication. That competitive advantage ends up working against SARS-CoV-2 when it comes to vaccines, making it a slower-moving target than other viruses.
“If this was HIV, for every variant we see now with SARS-2, we’d see 1,000 more,” Rutherford said.
Scientists were still cautious early on, said Mathai Mammen, Johnson & Johnson’s global head of pharmaceutical research and development, because while the repair mechanism for the coronavirus was known, the virus doesn’t stay as unchanging as measles, for example, where vaccination lasts a lifetime.
And the success rate for prototype vaccines is below 10%, Crotty noted. So it was unsurprising that vaccinologists, who deal with the hassles of proving out new immunizations in clinical trials, kept their hopes in check. And as expected, some of the vaccine candidates did face problems: Pharmaceutical giants GlaxoSmithKline and Sanofi delayed their vaccine to adjust its recipe in December, while Merck discontinued its vaccines a month later after disappointing early clinical trial results.
“I was always optimistic,” Deepta Bhattacharya, an immunologist at the University of Arizona, said. “This was an acute virus; you get it and get over it.” Viruses that cause chronic infections, like HIV and herpes, which the body doesn’t clear, are much harder to vaccinate against.
“Here, instead, we saw there was a natural pathway to immunization we could take advantage of,” Bhattacharya said.
A Sitting Duck
Coronavirus spikes, which the viruses use to attach to cells, also made vaccination easier. Early last year, scientists at the National Institutes of Health mapped the atomic structure of SARS-CoV-2. This launched an early campaign to design vaccines aimed against the spike, based on earlier work used to develop experimental SARS and MERS vaccines.
Blocking the spike made the most sense from an immunology standpoint, Akiko Iwasaki, an immunobiologist at Yale University, told BuzzFeed News. The spike is essentially the business end of the virus. If the spike is blocked from latching onto a cell, the virus is left powerless. So designing a vaccine that could summon antibodies aimed precisely at the spike protein (instead of somewhere else on the virus) seemed like a more direct approach to immunization.
There is a balance to this kind of specific targeting in vaccines. While all the most successful COVID-19 vaccines are aimed against the spike protein, some earlier prototypes targeted only its uppermost tip. That too-narrow approach proved to be less effective.
So did aiming too broadly. More-traditional vaccines, like the 50% effective Sinopharm vaccine made out of inactivated virus particles, train the body to make antibodies against the entire virus. That trick of biology explains its lower effectiveness.
“[The spike is] sort of a sitting duck in a lot of ways for vaccination,” Bhattacharya said. “It’s just asking for it.”
Built for mRNA
In particular, mRNA vaccines have emerged as the big winners in the pandemic, topping 90% effectiveness. These shots deliver genetic instructions to human cells so they make coronavirus spike proteins themselves. The proteins that the cells then churn out sensitize the immune system to spikes and thus protect it from a future infection by the genuine virus.
“These will be among the most efficacious vaccines that we have. Period,” said John Wherry, director of the Penn Institute for Immunology.
Two decades of research went into making mRNA vaccines that are perfect for battling coronaviruses, right down to figuring out the best kinds of fatty molecules to cushion the genetic material within each shot. “NIH did a great job at identifying exactly what part of the virus to use and exactly how to stabilize it in a form that makes the immune system recognize the most important part of it,” Wherry said.
That form is the “prefusion” stage of the spike protein, which is seen just at the moment where the spikes unfold and shape-shift to dock onto the cell surface. Interrupting this stage of an infection is like jamming gum into a keyhole, Wherry said. “You can stick gum all over a door and it might not do anything, but if you put gum in that keyhole, a key isn’t going to fit in there to open that door.”
Researchers knew about this vulnerability because of work done studying other viruses over the last decade. NIH scientists delivered a blueprint of this form of the spike protein to pharmaceutical companies so fast — within days of first seeing the coronavirus genome in January — that safety trials in people were already underway by April.
The payoff for all those decades of research came in November 2020, when late-stage clinical trials reported the 95% efficacy results for the Pfizer-BioNTech mRNA vaccine, which took aim at the SARS-CoV-2 prefusion spike.
“The way I think about it is simply that this organism was built with mRNA vaccination in mind,” said Musser, since the spike protein is such a solid target. For the flu, in contrast, many locations on the virus need blocking. “Imagine you need to target 20 locations on a pathogen, and each one only gives you 5% immunity,” Musser said. “You have to hit a whole lot of them.”
Even with such high levels of vaccine effectiveness, some immunized people may still get COVID-19, also known as a “breakthrough” infection. A small number of those people, drastically fewer than those who haven’t been vaccinated, can get severely ill. Of the more than 150 million people who have so far been fully vaccinated in the US, 439 have gotten so sick that they have died.
These breakthrough infections can help scientists understand more about “correlates of protection,” or precise measurements of the levels of antibodies and other immune cells needed to fend off the coronavirus. The problem is, in order to calculate this threshold, you need to take a blood sample just before a breakthrough infection occurs. Because the vaccines are so effective, this would require taking blood regularly from thousands of people just to catch the few infections that manage to break through.
“And it’s getting harder and harder to know as the number of infections go down,” Wherry said. “So we’re sort of victims of our own success in knowing how well we’re protected.”

To Boost, or Not to Boost
There are two big hanging questions about our COVID-19 vaccines: How long will immunity last? And will they be able to fight off new strains of the coronavirus?
On the first question, we really just don’t know for sure, because we have only been living with the virus for a year and a half. But some recent work has been encouraging; a May study published in Nature found that mild SARS-CoV-2 infections produced immune memory cells that last for at least a year, and possibly quite longer.
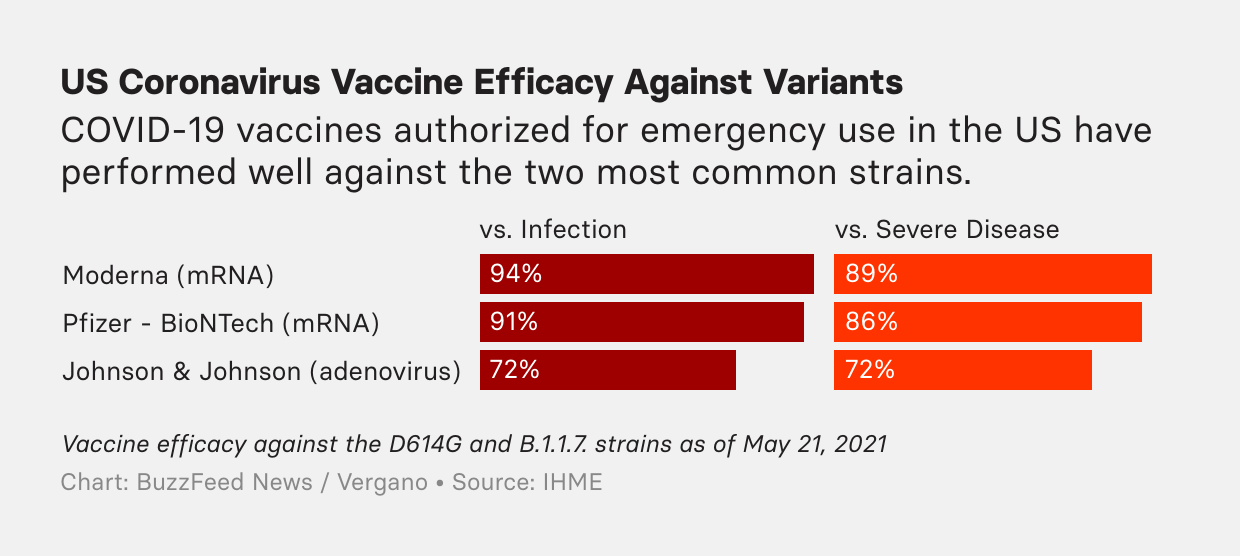
And of the CDC’s five listed coronavirus “variants of concern” that appear to be more threatening than the original SARS-CoV-2 strain, four of them reduce vaccine effectiveness — but not by very much. Even for the most worrisome variants — the P.1 variant, first observed in Brazil, and the B.1.351 variant, first identified in South Africa — the mRNA vaccines appear to be around 75% effective at preventing infections. Among the latest study findings, Public Health England reported last week that Pfizer’s mRNA vaccine is 88% effective against a highly contagious variant that is now widespread in India. AstraZeneca’s vaccine is 60% effective against the strain.
Nevertheless, pharmaceutical companies like Pfizer and Moderna are already testing booster shots trained against new strains of the virus, in case COVID-19 shots become a yearly recommendation, like seasonal flu shots.
Asked how soon he expects to see a booster, Fauci, at a May 25 White House briefing, said, “I'd like to give you a specific time and say ‘X number of months,’ but quite frankly, we don't know the answer to that right now.” Understanding when boosters might be needed will likely depend on figuring out the correlates of immunity, which he conceded might be years away, or, more likely, when doctors suddenly see lots of people sick either from a new variant or from immunity wearing off.
The bad news is, the evolution of new variants has accelerated, according to a recent review. “Selective pressure” on the coronavirus from vaccines, past infections, and treatments like monoclonal antibodies all require the virus to devise clever new ways to infect people.
Experts contacted by BuzzFeed News varied widely on their best estimates of when a booster shot might be needed. Some guessed next year, others said it could be another five years from now.
“I think we’re going to end up with a booster. The question is what the booster looks like,” Musser said. It might be a 50-50 mix of the original vaccine mixed with one targeted against a dangerous new variant. Or the booster might only be needed for older people who are at higher risk. Some people are inadvertently getting a booster because traveling to Europe requires a vaccination in the last six months, and they got their shots in December, Musser added. “That’s one way it will happen.”
While vaccines could slowly lose their efficacy against infections, they might still remain quite effective against severe disease. COVID-19 cases follow a two-part pattern: Early on, it’s contagious and causes cold symptoms; a week later it can turn into a serious illness. The more infectious variants are thought to evolve mostly in the earlier stages, when the symptoms are less severe.
That makes it harder to determine when booster shots should become a national imperative. “I’m probably still going to get the booster shot because I hate colds,” said Bhattacharya, even if the original vaccines still keep you safe from severe disease.
Scientists are also studying how well mRNA vaccines will tackle other viruses, like the flu, HIV, and the Nipah virus. The expectation is that viruses that are similar to SARS-CoV-2 — those with an essential, exposed target that doesn’t mutate much — will result in similar successes. But everyone wants to see the results of studies first, given past vaccine disappointments.
In the end, it’s a race between how quickly we can vaccinate the world and how quickly the virus continues to mutate. “It will be all about the organism,” Bhattacharya said. “That’s what always makes the decision.”
