The journalists at BuzzFeed News are proud to bring you trustworthy and relevant reporting about the coronavirus. To help keep this news free, become a member and sign up for our newsletter, Incoming.
Delighting public health experts, a second candidate coronavirus vaccine reported very effective test results on Monday, resetting expectations for when the deadly COVID-19 pandemic may end.
"The vaccines that we are talking about and vaccines to come are really the light at the end of the tunnel,” said Anthony Fauci, chief of the National Institute of Allergy and Infectious Diseases, during a Monday briefing on the vaccine made by the pharmaceutical company Moderna. “It is help that I’ve said is on the way.”
The promising new results from Moderna suggested that its vaccine is 94.5% effective at preventing COVID-19, coming just one week after the drug company Pfizer reported better than 90% effectiveness in its early data. Both firms say they will request emergency authorization from the FDA within weeks, kicking off a long-anticipated start of the campaign to vaccinate the United States.
But how soon will a coronavirus vaccine be available for you and your loved ones? Here’s what you need to know about the two leading vaccine candidates:
What do we know so far about how well these vaccines work?
So far, we have good news.
Both vaccines require two shots — spaced three weeks apart for Pfizer’s and four weeks apart for Moderna’s. To measure how effective a vaccine may be, both trials measure the difference in infection rates between participants who got vaccinated and those who received placebo shots.
Moderna said that among its 30,000 participants, 95 people were diagnosed with COVID-19 so far. Ninety of those cases were in the placebo group, while 5 had received the vaccine, leading to a 94.5% reduced risk of infection. Among the COVID-19 cases, 11 people had severe illness. All of those individuals were in the placebo group, suggesting that the vaccine is effective at blocking the worst disease symptoms.
Pfizer said that among its 43,500 study participants, 94 had been diagnosed with COVID-19. The company said on Monday, Nov. 9 that the difference between the two groups pointed to a greater than 90% effectiveness for the vaccine. The drug company did not release its breakdown of COVID-19 cases between the placebo and vaccinated groups, and it also did not release any information on the severity of symptoms in those who got sick.
These early analyses of the vaccine data were both determined by independent data review boards. Neither company has yet released its data in full.
Scientists said the findings were tremendously encouraging — and even surprising. The FDA set 50% effectiveness as its bar for allowing the use of the vaccines in its summer guidelines for vaccine makers, and Fauci had previously said he had hoped for 70% to 75% effectiveness.
“I said I would be satisfied with a 70% to 75% efficacy. Something like a 95% was aspirational,” Fauci said at the Monday briefing. “Well, our aspirations have been met, and that is very good news.”
"It’s remarkable actually," said Paul Offit, a vaccine expert at Children’s Hospital of Philadelphia, who sits on the FDA vaccine advisory committee.
Takeaways: 1) Proves Pfizer results not a fluke 2) Spike protein was right target 3) This'll make distribution easier (no need for special freezer) & faster (if all goes right, both companies will be churning out vaccine by early 2021) 4) "Normal" by late '21 is possible (2/2)
What do we know about how safe these vaccines are?
The best data on vaccine safety, so far, comes from smaller trials of hundreds of people conducted this summer, which found minor side effects, including sore arms, fatigue, and slight fevers, at a rate similar to that of other vaccinations. Because the great majority of severe side effects from vaccines come in the first two months after someone gets the shots, the FDA is requiring that companies collect two months of safety data from half the participants in their trials before they ask the agency to authorize their wider distribution. Moderna and Pfizer both said they expected to have that data available within weeks.
Typically, the FDA prefers to have two years of follow-up safety data before licensing a vaccine for wider use, but that would take too long in the midst of a deadly pandemic. Nevertheless, Moderna, Pfizer, and other vaccine makers have said they will follow people in their trials for signs of side effects even after the FDA authorizes them to distribute their vaccines. “I think we can be pretty confident they’re safe,” said Barry Bloom of Harvard’s T.H. Chan School of Public Health at a Monday briefing.
How soon will I be able to get a vaccine?
With the favorable results, Fauci and other experts have suggested that mass vaccination in the US can start around April.
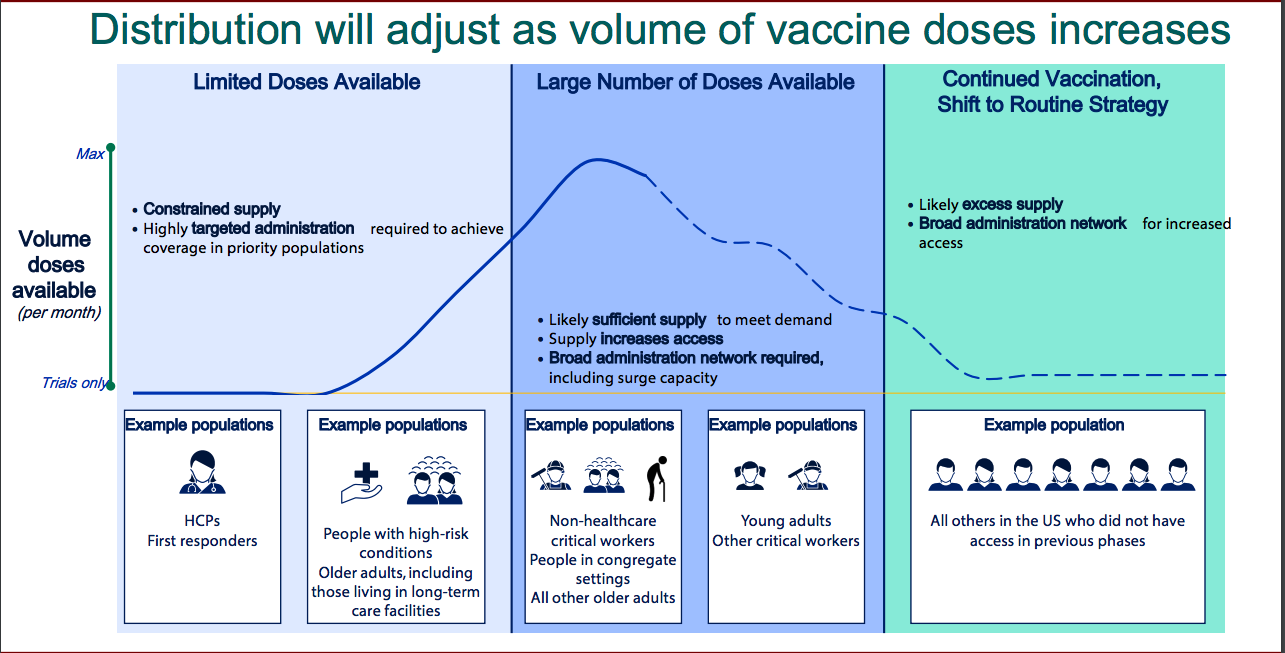
In public meetings, the CDC has outlined a three-phase vaccination campaign that starts after the FDA grants vaccine makers emergency authorization and the CDC’s advisory committee approves the nationwide distribution strategy for each.
The plan for every state and jurisdiction varies, but all of them will first give shots to healthcare workers, about 20 million people nationwide. Tremendously vulnerable, doctors, nurses, and other medical workers keep hospitals from collapsing in the face of surging caseloads. More than 215,000 healthcare workers in the US have been diagnosed with COVID-19.
“If we don’t protect the healthcare workers at every level … we’re not going to have a healthcare system,” said Bloom.
Healthcare workers will likely start receiving shots in December. The first six weeks of the rollout will see small but increasing numbers of doses aimed at these essential personnel, the CDC’s Amanda Cohn said at an Advisory Committee on Immunization Practices meeting in October.
Moderna said it has nearly 20 million vaccine doses ready to ship in the US by the end of 2020 and will produce up to 1 billion doses worldwide in 2021. Pfizer projected that it will manufacture 50 million vaccine doses in 2020 and as many as 1.3 billion doses in 2021 to be distributed worldwide.
Many states also prioritize older people, essential workers, and people with illnesses, such as diabetes and heart disease, that put them at a higher risk of dying. Nursing homes are a particular focus, where more than 65,000 people have died from COVID-19. These vulnerable populations make up more than 80 million people, nearly a quarter of the total US population.
The next phases will prioritize other older adults, young adults, and then the general public over the course of 2021. Following the promising new data, Fauci has stated that could come as early as April next year.
How long will the vaccines protect people from COVID-19?
We don’t know. Researchers are still following participants from the earlier safety trials to see if their levels of antibodies and other cells that fight the coronavirus drop precipitously or remain steady, which would suggest that the vaccine could protect you for a longer period of time.
However, time has to pass to measure this. The coronavirus has only been spreading among humans for less than a year, and the vaccine trials have only been underway for months.
“We do not know at this point what the durability of protection will be, and that’s the reason why we have long-term follow-ups,” Fauci said on Monday.
One yardstick for a long-term protective effect from the vaccines will be comparing vaccine-induced immune responses to those seen in patients who have recovered from COVID-19, Bloom said. If vaccinated people retain more of an immune response than recovered patients, that would point to long-term immunity. Otherwise, it might mean we will need to have a yearly coronavirus vaccine, just like the flu shot.
How will these vaccines get distributed across the country?

There are at least three big challenges with distributing the vaccines: The first is the need to manage the “cold chain” requirements that keep vaccines below freezing temperatures for long-term storage. The second is the money to have states distribute the vaccine, now held up in Congress in a larger disagreement over a second federal coronavirus aid package for businesses and people who are unemployed. The last, and hardest, roadblock is “vaccine hesitancy”: Polls suggest that only about half the US population is now willing to get the vaccine, down from three-quarters in the spring, according to surveys from the Pew Research Center.
The Pfizer vaccine requires long-term storage at -94 degrees and will need to be distributed in boxes lined with dry ice containing thousands of doses. Rural hospitals have already said they can’t afford the freezers needed to store the vaccine, which may be better suited for large medical centers and big cities. At a Council of Supply Chain Management Professionals panel last week, Angela Watson, UPS’s vice president of global healthcare sales, said the delivery company was readying its own dry ice production, in anticipation of keeping such ultra-cold vaccines viable.
The Moderna data takes some pressure off distribution, however. The biotech firm announced on Monday morning that its vaccine candidate can stay in a refrigerator for a month and in a standard freezer at -4 degrees for six months. That makes the vaccine easy to use for pharmacies, medical practices, and clinics, where most shots are now given, and eases distribution in rural states.
If temperature conditions aren’t exactly right, vaccine “spoilage” is a real worry, experts at the supply chain meeting said, with perhaps half of all flu vaccines exceeding their “use by” dates. (The CDC, which oversees flu vaccine distribution in the US, told BuzzFeed News it could not confirm that number.)
How many other vaccine candidates are there?
Worldwide, 12 vaccines are in late-stage trials, with four frontrunners, including Moderna's, sponsored by the federally funded Operation Warp Speed, which is paying for manufacturing plants ahead of the vaccine proving effective. Pfizer has a $1.95 billion advance purchase agreement with OWS to distribute the vaccine in the US but hasn’t accepted development money from the US government.
The next two OWS vaccines expected to report results are from Janssen Pharmaceuticals and the University of Oxford’s partnership with AstraZeneca. Both of those trials had been briefly halted to investigate whether patients had developed illnesses linked to the vaccines, resuming after public health agencies investigated and determined that the vaccines likely played no role. The Janssen vaccine might be the most intriguing because it is a single-shot vaccine, unlike the other frontrunners, cutting down the need for tracking recipients and having them return in time to receive their second shot.
How are the Pfizer and Moderna vaccines different from the rest?
The Moderna and Pfizer vaccines are both messenger RNA (mRNA) gene vaccines, a kind never before approved for use in people. They carry genes that instruct cells to make viral proteins from the “spikes” that SARS-CoV-2 uses to invade cells, priming the body's immune response to attack the virus. Easily manufactured, these genes are packed in fatty molecules and injected straight into the body. Traditional vaccines require growing batches of a virus — a long and delicate process — and then killing them and injecting the dead virus to trigger an immune response.
The Janssen and AstraZeneca vaccines are also a newer approach, in which a harmless cold virus is genetically engineered to contain the genes for coronavirus spike proteins. These combinations are injected as a vaccine to similarly trigger an immune response to the coronavirus.
If the vaccines are authorized, will life go back to normal?

No. An emergency use authorization may come from the FDA as soon as next month, but people will still have to wear masks, socially distance, and frequently wash their hands to quell the spread of the coronavirus throughout 2021. Importantly, this also applies to people who have been vaccinated — because no vaccine is 100% effective, Bloom noted. ”If all the vaccines get up to 90% effective, that’s terrific, but we will still need around 700 million doses for everyone,” he said, noting that the Moderna and Pfizer vaccines require two doses each.
Estimates vary on how much of the population needs to either get vaccinated or recover from COVID-19 to blunt its spread, also known as herd immunity. One frequently used estimate suggests that 60% of people will need some form of immune protection — which would total 192 million people in the US alone. And that ignores the wider spread of the virus worldwide, with a global population of 7.8 billion needing vaccination in order to prevent cases from spurring outbreaks in unvaccinated pockets of the population through travel. Though the measles vaccine offers a lifetime of protection, outbreaks have occurred in recent years as the virus spreads among unvaccinated portions of the population.
On Monday, Fauci cautioned that the good vaccine news should help motivate people to stay vigilant with social distancing throughout the Thanksgiving and Christmas holidays.
“We are now still in a critical and dangerous time,” said Adm. Brett Giroir of the Department of Health and Human Services at a COVID-19 briefing on Monday. “We need to double down on protective measures. And if we do, we can save thousands of lives.”
