BuzzFeed News has reporters around the world bringing you trustworthy stories about the impact of the coronavirus. To help keep this news free, become a member.
On January 17, the world’s most trusted public health agency, the Centers for Disease Control and Prevention, announced it was screening travelers from Wuhan, China, because of a new infectious respiratory illness striking that city.
It was the CDC’s first public briefing on the outbreak, coming as China reported 45 cases of the illness and two deaths linked to a seafood and meat market in Wuhan. Chinese health officials had not yet confirmed that the new illness was transmitted from person to person. But there was reason to believe that it might be: four days earlier, officials in Thailand confirmed their first case, a traveler from Wuhan who had not visited the seafood market.
“Based on the information that CDC has today, we believe the current risk from this virus to the general public is low,” said Nancy Messonnier, the CDC’s director of the National Center for Immunization and Respiratory Diseases. Messonnier, 54, was a veteran of the CDC’s renowned Epidemiological Intelligence Service, where she had risen through the ranks during the national responses to the anthrax attacks and the previous decade’s swine flu pandemic to eventually head the agency’s vaccines center.
Most of the novel coronavirus’s infections apparently went “from animals to people,” she explained, and human transmission was “limited.”
There were many reasons why the information the CDC had on January 17 was wrong. It was wrong because China’s leaders withheld what they already knew about the virus from the World Health Organization. It was wrong, perhaps, because Trump administration officials had cut CDC staffers in Beijing who might have reported the truth directly from China. And it was wrong because past coronavirus outbreaks provided a false guide to an illness new to humanity.
That last reason — a fateful misjudgment of the basic biology of the virus — drove a flawed strategy to contain the outbreak. In 17 press briefings from January to March, the agency pushed the idea that if travelers, first from Wuhan and then from China, were quickly identified, traced, and isolated, it could “slow and reduce” the spread of the virus on US soil. Believing people without symptoms didn't spread the virus, the agency limited testing, discouraged masking, and left the country blinded by a faulty COVID-19 test.
Then, when the containment strategy's failure became undeniable, just one day before the WHO declared the outbreak a pandemic — the CDC disappeared from public view.
“When the outbreak started, we had an aggressive tracing program, but unfortunately, as the cases rose, it went beyond the capacity,” CDC Director Robert Redfield later testified to Congress.
“We lost the containment edge.”
More than 120,000 US deaths and counting later, public health experts disagree whether the CDC — which only resumed its briefings on the coronavirus in June after its three-month vanishing act — could have ever contained SARS-CoV-2. Everyone agrees, though, that the US response has been a disaster across the federal government, with the CDC the most visible face of failure.
“We certainly could have done enormously better than we did,” former CDC director Tom Frieden told BuzzFeed News.
Could the US have stopped the outbreak completely, like Taiwan and New Zealand have done?
“Probably not,” said Frieden. “Could we be Germany, with way, way fewer cases and deaths and less economic dislocation? Absolutely.”
A fatal misunderstanding about the new virus
At that first CDC coronavirus briefing, Messonnier explained what two other coronaviruses, MERS and SARS, could tell us about how to contain the mysterious new virus. And she described the crux of their containment plan: stopping the disease from entering the US by screening for travelers with a fever and a cough, testing them, and putting sick people in isolation.
“We know from investigation of those two viruses that they are more likely to spread when somebody is more contagious,” she said. “Asymptomatic people can spread, but at a much lower rate.”
SARS was a novel coronavirus whose outbreak in 2002 and 2003 killed 774 people, largely travelers and hospital personnel. It was most contagious when someone was severely ill, making quarantines very effective because by the time someone was most contagious, they were usually already confined to a hospital bed. MERS, which has killed at least 845 people since 2012, is likewise most often transmitted from terribly ill patients.

SARS-CoV-2 is entirely different. We now know that COVID-19 patients have the heaviest viral load — and appear most infectious — at the onset of symptoms, not at the end. People who haven’t yet developed any symptoms, and therefore wouldn’t show up in the CDC’s temperature checks, are responsible for more than 40% of the virus’s spread. And only about 45% of cases early in an infection will develop a fever.
In its first briefing, CDC officials acknowledged they still knew very little about the new virus. “CDC will adjust its screening and response procedures appropriately,” said the CDC’s Marty Cetron, director of the Global Migration and Quarantine Division.
Three days later, China confirmed the virus spread from person to person. Within a week, Chinese officials took the unprecedented move of quarantining Wuhan, a city of 11 million people, sending a clear message that the illness could readily spread outside the hospital setting.
Nevertheless, the CDC stuck to its screening plan.
“We are looking for returning travelers who have fever, cough, and respiratory symptoms,” said Messonnier at the agency’s next briefing on January 23. At the following briefing, on January 27, she reiterated the core assumption of the agency’s screening strategy: “We at CDC don’t have any clear evidence of patients being infectious before symptom onset.”
We now know that the coronavirus was likely already spreading on US soil by this time, with at least four lines of evidence supporting that conclusion. There is the case of a Northern California woman who became sick on January 31 and died within a week. And there is genetic evidence: The slow but steady mutation rate of the novel coronavirus, amounting to a few tiny changes every month, serves as a kind of footprint to track its spread. Scientists currently believe that trail points to the virus’s spread starting in November in China and arriving in Washington state in January, spreading through undetected, asymptomatic cases.
On January 30, with 7,800 known cases in 22 countries, the World Health Organization declared the outbreak a Public Health Emergency of International Concern. The same day, the US announced the first case of person-to-person transmission of the virus in the country; the husband of a Chicago woman who had acquired the illness in China.
The next day, Health and Human Service Secretary Alex Azar announced the outbreak was a national public health emergency, and the White House declared a ban on all foreign travelers from China.
China typically sent 14,000 air passengers a day into the US. The order allowed the quarantine of anyone exposed to the virus, and both Redfield and Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, cited the risk of asymptomatic transmission to justify the action.

Among public health experts, the travel ban was divisive. Some called it both an overreaction and too late, while others have said that such restrictions applied worldwide delayed the outbreak of the virus from China until mid-February.
The real problem was that the travel bans by themselves were a Band-Aid on a dam about to burst, only helpful when paired with public health agencies performing widespread testing and isolating infected patients — both with and without symptoms.
That didn’t happen. California Department of Public Health officials would later complain in a CDC Monthly Morbidity and Mortality Report that the travel restrictions and screening were a distraction and a drain on its resources. “Monitoring travelers was labor-intensive and limited by incomplete information, volume of travelers, and potential for asymptomatic transmission,” said the report. “Despite intensive effort, the traveler screening system did not effectively prevent introduction of COVID-19 into California.”
The real problem was that the travel bans by themselves were a Band-Aid on a dam about to burst.
At a January 31 CDC briefing, Messonnier announced that 195 passengers brought back from Wuhan would be involuntarily held for 14 days at a California air base, the first time in over 50 years that the agency had issued a quarantine order. “We are preparing as if this were the next pandemic, but we are hopeful still that this is not and will not be the case,” she said.
Meissonier’s “next pandemic” statement should have triggered action across the federal government, Frieden, the former CDC director, told BuzzFeed News.
“That's the point at which Iceland and Korea and Germany and a bunch of other countries got the private sector commercial laboratories into scaling up test production, ventilator production, respirators, protective equipment production,” he said.
But that didn’t happen. The next two months would make this failure clear.
“You would have had to test everybody who comes to the emergency room”
As the virus began to spread in the US, a dire lack of testing defined the month of February, driven by mistakes at the health agency.
The CDC began February by sending states instructions on diagnosing and testing people for the novel coronavirus. The “Patients Under Investigation” criteria “have been developed based on available information about this novel virus, as well as what is known about SARS and MERS,” the agency told state health officials on February 1.
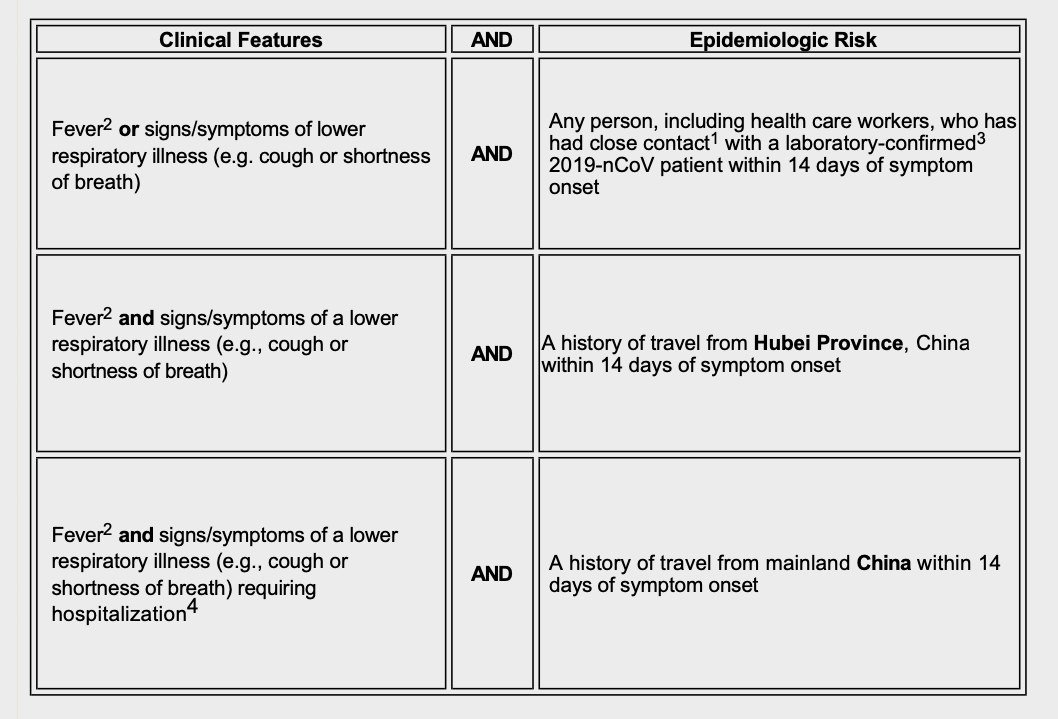
Despite the need for widespread testing made apparent by the reports of people without symptoms spreading the disease, the CDC’s criteria was very narrow: Only people with a fever, cough, and shortness of breath who had traveled to Hubei province or had close contact with a confirmed patient could be tested. Hospitalized patients with those symptoms who had traveled to China within the last 14 days could also get a test.
Driven in part by these restrictions, others with symptoms — including healthcare workers concerned about their exposure to the virus in hospitals — initially struggled to get tested.
Genetic tests for coronavirus work by detecting traces of signature genetic material from the virus called RNA. The CDC developed a test that looked for three pieces of RNA found in the novel coronavirus. The WHO, meanwhile, had been sending a reliable test made by the German Center for Infection Research worldwide since the middle of January. China had five commercial tests already on the market by the beginning of February.
The US had only the CDC test, used on fewer than 250 people by February 1 and processed solely at the agency’s Atlanta headquarters. When asked about the other testing options, Messonnier said the CDC preferred to have its own, more precise test.
Whatever the reason, on February 5, Messonnier touted the authorization of its genetic test for wider use alongside FDA Chief Scientist Denise Hinton, who said her agency had sent the template for similar authorizations to 35 other test developers.
Things went bad quickly. On February 9, the Association of Public Health Laboratories sent a message to its member labs in all 50 states, notifying them that they’d had problems verifying the test.
In a press conference five days later, optimism about containing the virus began to fade, with Messonnier acknowledging “problems with one of the reagents” in the diagnostic test. “We must prepare for the possibility that at some point we may see sustained community spread in other countries or in the US, and this will trigger a change in our response strategies.”
Asked what that meant, Messonier replied that switching from containment to “mitigation” of the outbreak would mean “social distancing, school closures, canceling mass gatherings,” as well as “teleschooling, telemedicine, teleworking” — the first mention of such measures at a CDC briefing.
But following the CDC’s playbook on flu pandemics, Messonnier discouraged the public from using face masks, echoing similar advice from the WHO and US Surgeon General Jerome Adams. Since masks were thought to prevent the transmission of a virus from coughing, health officials feared a run on surgical masks would keep them from reaching vulnerable public health workers in hospitals, where most infectious patients driving a coronavirus outbreak would be located.
In April, facing increasing pressure from public health experts, the CDC urged Americans to don cloth masks, citing the asymptomatic transmission risk first noted by Redfield and Fauci at the end of January. But the damage was done: Months later, the initial confusion over the utility of widespread use of face coverings has only made masks more of a political flashpoint.
Meanwhile, as February came to a close, the CDC’s test still wasn’t working in most labs. An FDA official arrived to inspect the CDC’s lab on February 22, a Saturday, but was not allowed in until the next day, where he reportedly found signs of contamination fouling the test. A June HHS investigation confirmed that the CDC’s Respiratory Virus Diagnostic Lab, which was assembling the reagents, was also likely contaminated. The investigation noted that the agency skipped quality control testing on the kits before shipping them to public health labs.
Frustrated, the Association of Public Health Laboratories on February 25 requested that the FDA allow its labs across the country to go ahead with their own tests. Only a dozen labs were able to run the CDC’s three-primer test at the time, and the federal health agency was running tests for only around 100 patients a day.
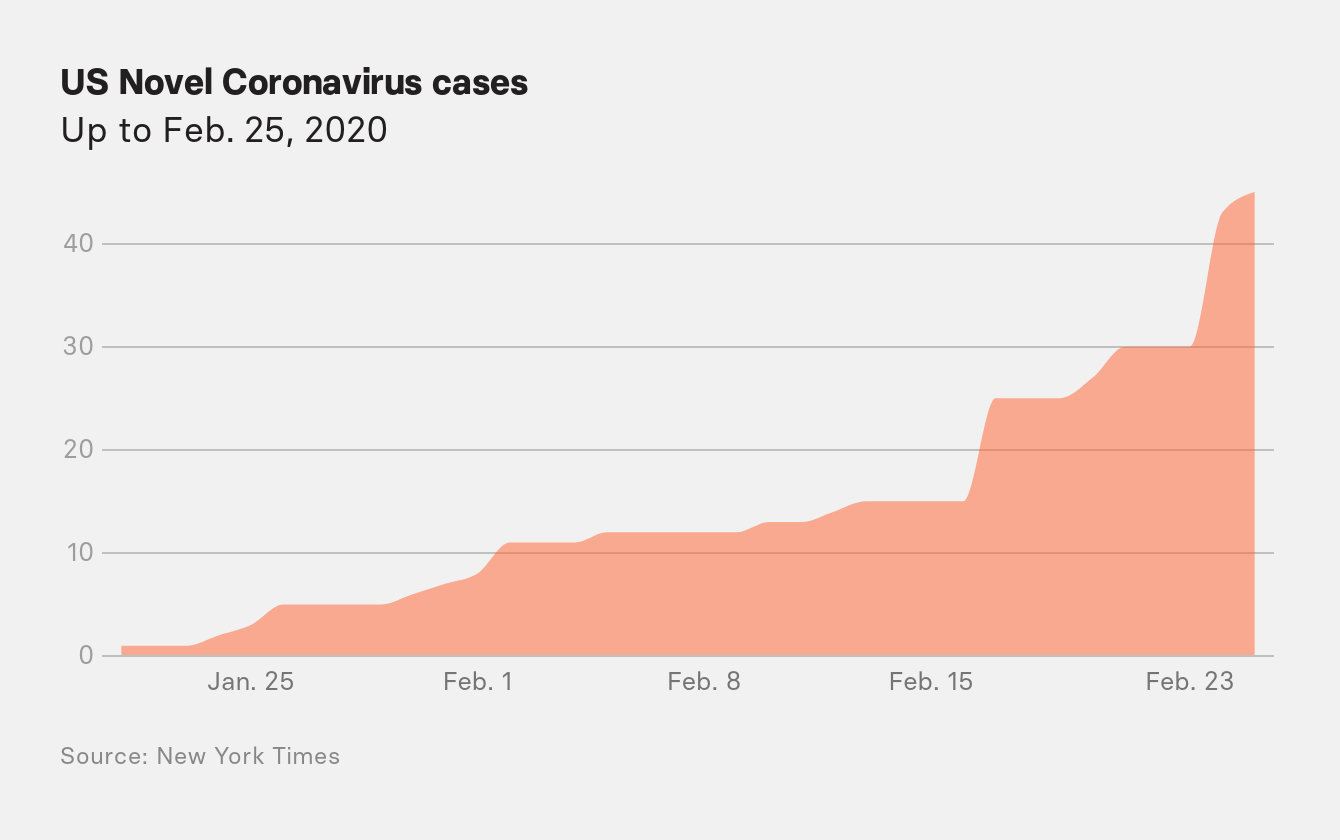
Do you remember on Feb 25 when @DrNancyM_CDC said "disruption to everyday life might be severe"? I think about that all the time.
In her opening remarks at a fateful CDC briefing on February 25, Messonnier finally dropped any pretense that the US would not suffer from the outbreak. “You should ask your children’s school about their plans for school dismissals or school closures,” she said. “You should think about what you would do for childcare if schools or daycares close, if teleworking is an option for you.”
The remarks set off a news firestorm, which, combined with a stock market drop, reportedly enraged President Donald Trump. The next day, he replaced Azar with Vice President Mike Pence as chair of his coronavirus task force, which began daily briefings that would soon replace the CDC’s. "It's going to disappear. One day, it’s like a miracle, it will disappear," Trump said on February 27, at a Black History Month reception.
The FDA and the CDC soon gave public health labs, starting with New York state’s, permission to use their own tests instead of the CDC’s. No other authorized tests were available yet for the virus in the US at the time, despite the FDA’s claim that 35 companies were working to develop their own tests at the beginning of the month.
Then, weeks after its first guidance was issued, the CDC broadened its criteria for testing patients, eliminating the travel requirement and allowing anyone with pneumonia to get tested. One day later, Washington State Department of Health officials reported a teenager had tested positive for the coronavirus without any link to a travel-connected case, making it the first known case of community spread in the country.
In an interview with BuzzFeed News, the CDC pushed back against the notion that the health agency could have detected the spread of the virus earlier if it had not had such narrow testing guidelines and a defective test. The CDC would have needed “a huge amount of testing available at that time” to detect the virus before it had started spreading on US soil, said Greg Armstrong, a member of the agency’s COVID-19 Response Team who later worked on a report finding genetic signs of the coronavirus in the US since late January.
“It still would have been difficult to pick up community transmission in early February. I think probably at best we would have been able to pick it up a week or two earlier,” Armstrong said.
“You would have had to test everybody who comes to the emergency room,” he added.
But many experts have noted that this level of widespread testing was a challenge, not an impossibility. “The federal government has consistently underestimated the need for urgency around testing for this virus,” said Boston University School of Public Health Dean Sandro Galea, adding that this misjudgment has been a fiasco for public health labs.
Two days later, on February 29, those same Washington state officials reported the first suspected US death from COVID-19, of a man in his fifties with an underlying illness. They also reported the first outbreak in a nursing home, the Life Care Center of Kirkland, Washington, where ultimately at least 43 residents died. The same day, Gov. Jay Inslee declared a state of emergency.
The virus was clearly loose in the US.
Asked on the day of the Life Care Center report whether nursing homes should be closed, Messonnier replied, “Right now, we still judge the general risk to the American public to be low, and that includes residents of long-term care facilities.”
Another Washington state resident, a man in his seventies, died that Sunday. And on Monday, four more died, bringing the death total in the US to six people. Four of them were Life Care Center residents. Nursing home residents would eventually account for about 40% of coronavirus deaths in the US.
At the same CDC briefing on February 29, Jeff Duchin of Seattle and King County’s public health office revealed that the man who had died was only eligible for testing because of the CDC’s updated recommendations.
“If we had the ability to test earlier, I’m sure we would have identified patients earlier in the community,” he said.
“It wasn't impossible to contain.”
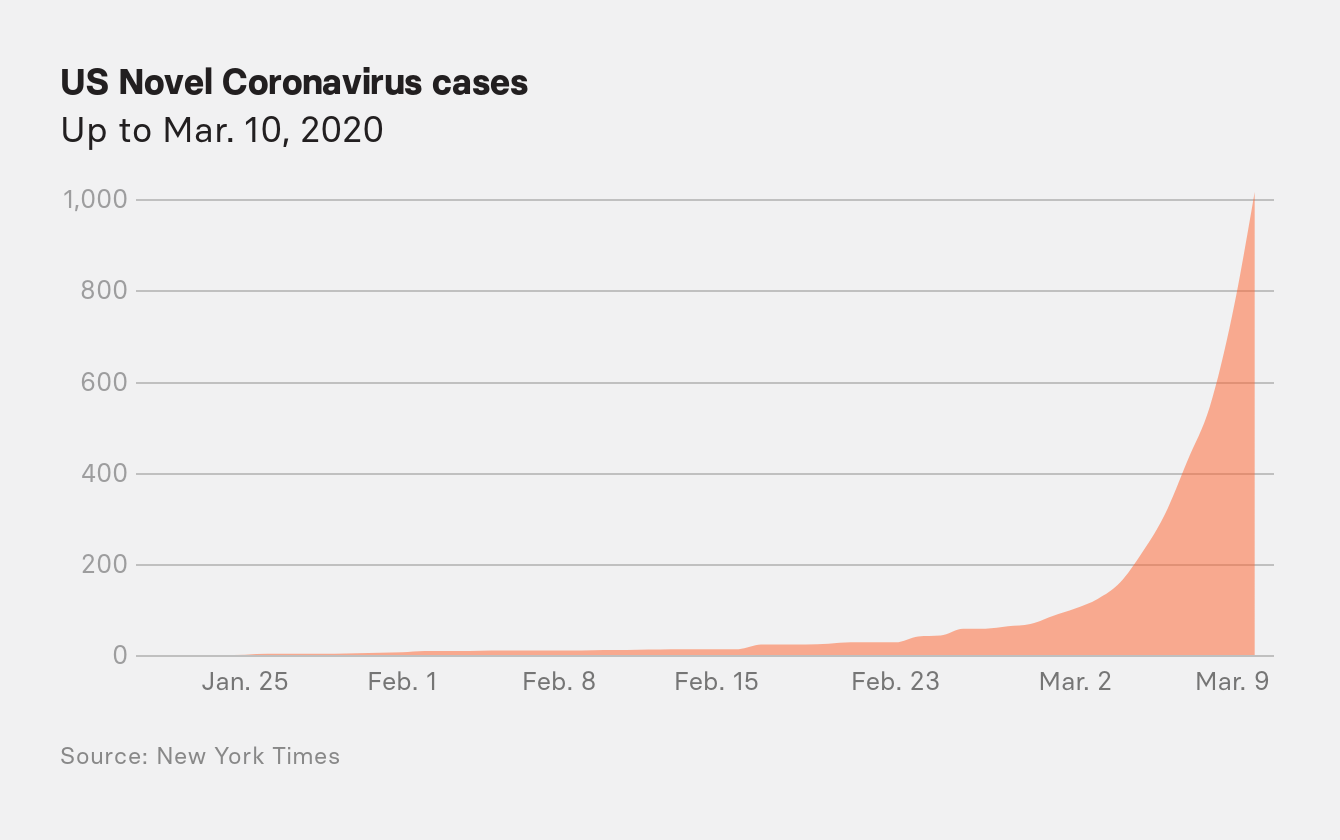
Nationwide, however, the loosening of the CDC criteria mattered very little as March began; there still weren’t enough tests to go around.
At a White House briefing on March 3, FDA Commissioner Stephen Hahn promised that a private lab, Integrated DNA Technologies, would soon pick up the slack by performing up to 1 million of the CDC-style tests by the end of the week. Pence would back away from this promise a few days later. The FDA would not approve the first private test under emergency authorization until March 12, and physicians would be reporting widespread testing shortfalls through April. Many states to this day are still not performing enough tests.
Messonnier put the blame on doctors. “CDC’s criteria for patients under investigation has always started with the importance of astute clinicians who are making judgments about what their patients are likely to have,” she said on March 3. “So we’ve always allowed those patients to be part of the testing criteria.”
With testing restrictions loosened, US case numbers suddenly started to mount into the hundreds, climbing steadily day by day. On March 10, Messonnier reported that the US had more than 500 confirmed cases. “This virus is capable of spreading easily and sustainably from person to person based on the available data,” she said.
The assessment was a complete reversal of the information the agency had at its first briefing less than two months earlier. This would also be the last time the CDC addressed the public until June.
The next morning, on March 11, the WHO officially declared the global spread of the coronavirus a pandemic. In a few days, US cases would begin to double every few days, commencing a dreadful exponential rise in cases and deaths that would ravage New York City.
Trump himself only acknowledged the virus was “not under control” on March 16, an admission bookended by groundless reassurances that the virus would go away. As the White House’s coronavirus task force replaced the CDC as the main information source for a fearful public, Trump’s increasingly erratic messages — such as his calls for treatment with an unvetted malaria drug, his refusal to wear a mask, and his attacks on scientists — politicized a nationwide health crisis.
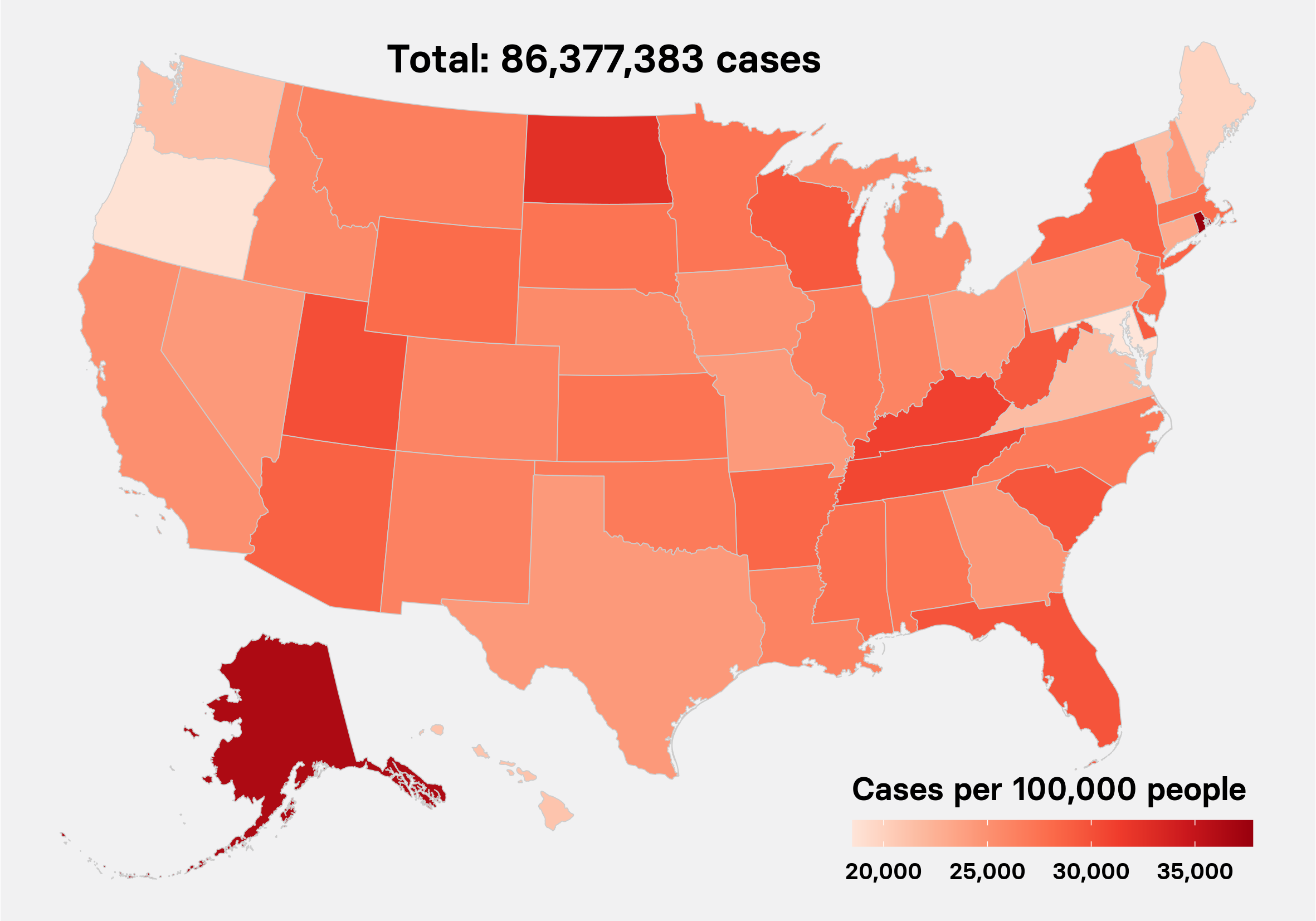
New confirmed cases in the US by day
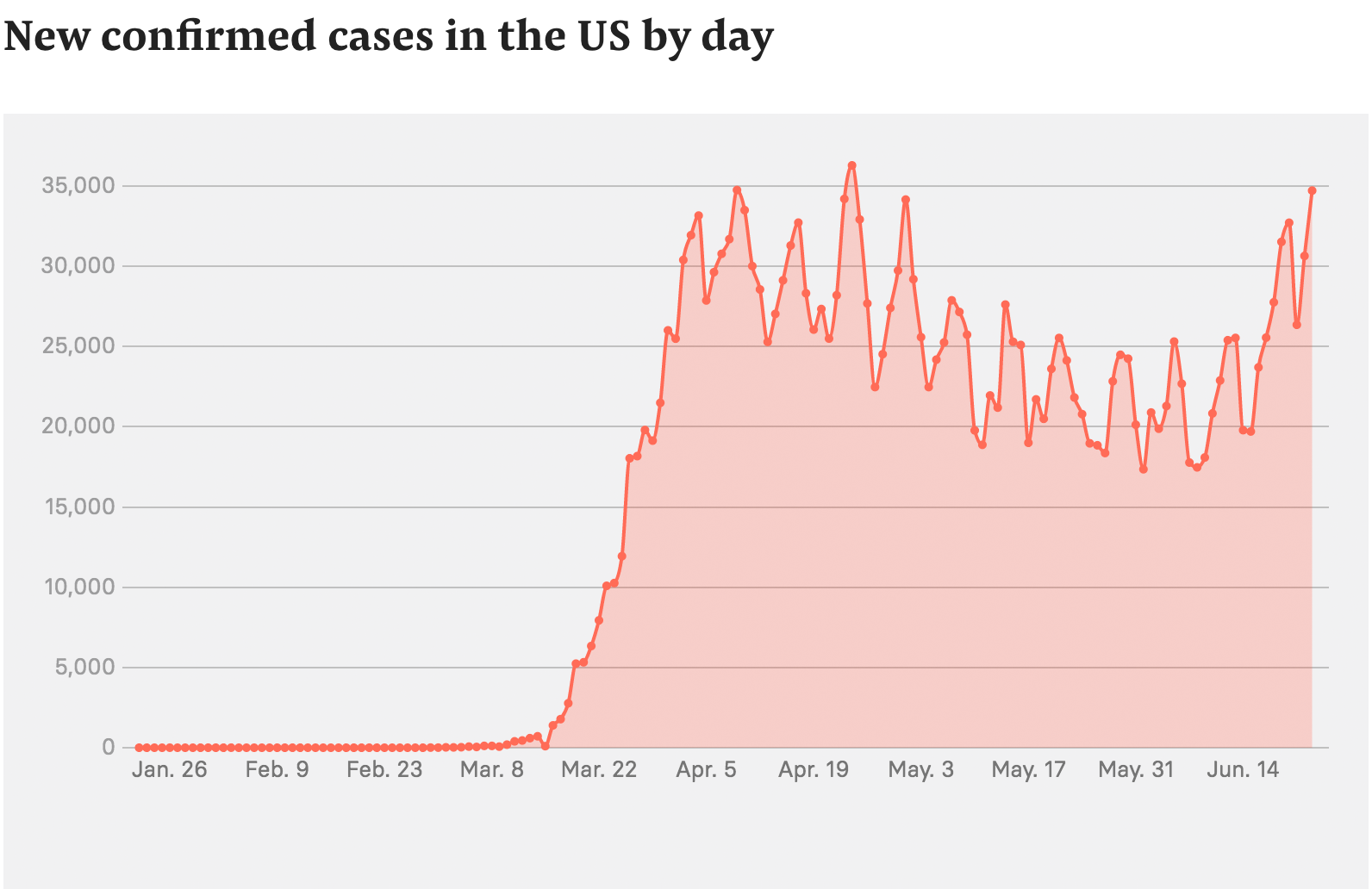
Since March, as case counts climbed to more than 2 million, blame for the US’s failure to keep the coronavirus contained — when countries like South Korea, Germany, and even China succeeded — has extended far beyond the CDC.
The Trump administration has blamed China for covering up its early cases, and the WHO for being too compliant with China to get such information out. A whistleblower complaint filed by Rick Bright, the former director of the Biomedical Advanced Research and Development Authority, paints a picture of White House officials uninterested in ramping up the production of medical equipment early in the outbreak. Azar was blamed for not coordinating efforts at the CDC and the FDA, leaving the botched test the only one available, and for overselling his agency’s efforts to Trump. Some news accounts blame the White House economic team for pushing back against medical and bioterror advisers who wanted travel bans sooner, and against replenishing the national stockpile of protective medical equipment in January. Trump’s son-in-law, Jared Kushner, was put in charge of acquiring medical equipment, an effort described as “fumbling” and “inexperienced” that became the subject of another whistleblower complaint. Feeling mounting pressure, the White House has most recently taken aim at the CDC.
Nevertheless, the CDC’s failures to dump its botched test, expand its testing criteria, and rethink a virus that spreads without symptoms, has baffled experts.

“One of the things I find so puzzling is you see mistake after mistake after mistake, one right another,” said Harvard global health professor Ashish Jha. “My sense is that all of these people knew things were going very badly. But none of them had the courage to speak up at the White House and push for an adequate federal response.
“Sure, the biology of the virus made it harder; this isn’t SARS,” he added. “But it wasn't impossible to contain.”
“Sure, the biology of the virus made it harder; this isn’t SARS. But it wasn't impossible to contain.”
Frieden, the former CDC chief, argued that the agency failed largely because it has been shackled to an inept administration. “The overarching problem has been the failure to have a clear national strategy, a national plan, and consistent communication. Everything really stems from that,” he said.
“Hindsight really always is twenty-twenty, and there were absolutely mistakes made by CDC, but also by FDA, by HHS — and by the White House.”
On June 12, after more than 113,000 Americans had died in the pandemic, the CDC held its first briefing in three months. In a presentation by CDC Director Redfield, he shared the agency’s recommendations for lowering the risk of infection as states begin to reopen.
Along with the CDC’s Jay Butler, deputy director for infectious diseases, he emphasized staying 6 feet apart, washing hands, and wearing cloth masks. The pair deflected questions on whether the guidelines applied to mass rallies announced by the president’s reelection campaign. Asked about the surges in cases currently hitting states like Arizona, Florida, and Texas, Butler suggested it could be explained by a rise in testing, a suggestion Fauci would dismiss in a congressional hearing two weeks later. Messonnier was not on the call.
“You know, I'm hopeful that we'll continue to have these dialogues,” said Redfield at the close of the briefing. ”I want to thank you for what you do to help communicate the messages that we feel are important to get to the American public.” ●