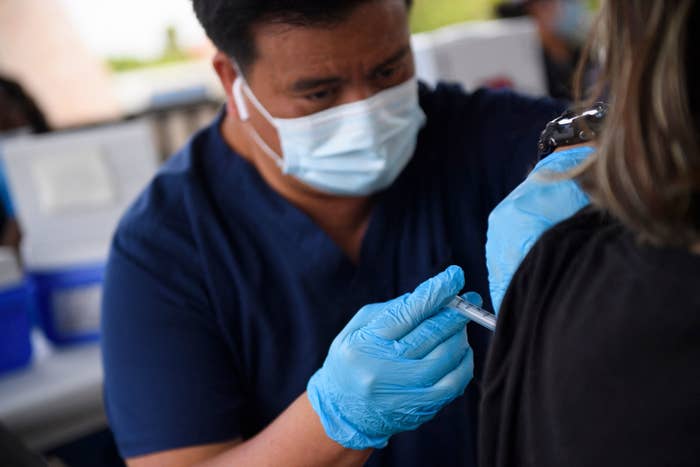
The FDA authorized a third COVID-19 vaccine dose for severely immunocompromised people on Thursday night, acknowledging the threat posed by the current surge to those at serious risk of contracting the virus.
The announcement greenlit a booster Moderna or Pfizer vaccine dose for organ transplant recipients, who often take drugs to suppress their immune systems and prevent their bodies from rejecting a transplanted organ. It also authorized third doses for the small number of people with conditions that cause an equivalent level of vulnerability to their immune systems.
"The country has entered yet another wave of the COVID-19 pandemic, and the FDA is especially cognizant that immunocompromised people are particularly at risk for severe disease," acting FDA Commissioner Janet Woodcock said. "Today’s action allows doctors to boost immunity in certain immunocompromised individuals who need extra protection from COVID-19."
Other fully vaccinated people do not require a third dose at this time, the agency said.
Researchers have recently reported that two doses of an mRNA vaccine do not produce the same high level of antibodies in severely immunocompromised people as they do in healthy individuals. Public health experts recommend that immunocompromised people continue to wear masks and stay socially distant.
A study published in the Journal of the American Medical Association in May this year detected antibodies in 54% of transplant recipients after the second vaccine shot, but it was "still well below what's typically seen in people with healthy immune systems," said Brian Boyarsky, the lead researcher and surgery resident at the Johns Hopkins University School of Medicine.
The FDA's announcement follows a CDC vaccine safety panel last month urging the change, over concerns that some people with cancer and those with organ transplants might be insufficiently protected by just two shots of the vaccines. A recent New England Journal of Medicine report confirmed that a booster raised antibodies in those people to levels seen in healthy people who had gotten two shots.
Less than 3% of US adults fall into categories now recommended for the booster, and all authorized US vaccines still look very effective against severe Delta variant infections in healthy people, according to National Institute of Allergy and Infectious Diseases chief Anthony Fauci, speaking Thursday at a White House briefing.
CDC Director Rochelle Walenskyis said in a statement that the booster recommendation is "an important step in ensuring everyone, including those most vulnerable to COVID-19, can get as much protection as possible from COVID-19 vaccination."
The US has seen a massive surge in new cases and hospitalizations amid the spread of the highly contagious Delta variant and the loosening of safety measures. In late July, in a reversal of its previous guidance, the CDC recommended vaccinated people mask up in places with high infection rates. And as schools reopen, thousands of students have already been forced to quarantine after being exposed to COVID-19, especially in parts of the country with the lowest vaccination rates.
Though officials have largely resisted bringing back restrictions, some cities are requiring proof of vaccination for most indoor activities, while California has become the first state to mandate vaccines for certain workers or subject them to weekly testing.
Only 50.4% of the US population is fully vaccinated, and 59.2% have received at least one dose of the vaccine.

