The Ohio Department of Corrections intended to illegally import drugs for executions, according to an FDA letter obtained by BuzzFeed News. Ohio is now the second known state to have moved toward buying sodium thiopental overseas — the other being Nebraska.
In a June letter, the FDA wrote to Ohio, warning the state that importing the drugs would be illegal.
"Please note that there is no FDA approved application for sodium thiopental, and it is illegal to import an unapproved new drug into the United States," wrote Domenic Veneziano, the director of FDA's import operation.
The letter, which was sent to the head of the Ohio Department of Rehabilitation and Correction, was obtained through a Freedom of Information Act request. A department spokesperson did not respond when asked if the state still intends to import the drug.

In similar letters to Nebraska and its Indian drug supplier, the FDA cited "media reports" as the source of its information that the state was attempting to import execution drugs. However, the letter to Ohio cited only "information received by the agency." The FDA declined to specify how officials were tipped off.
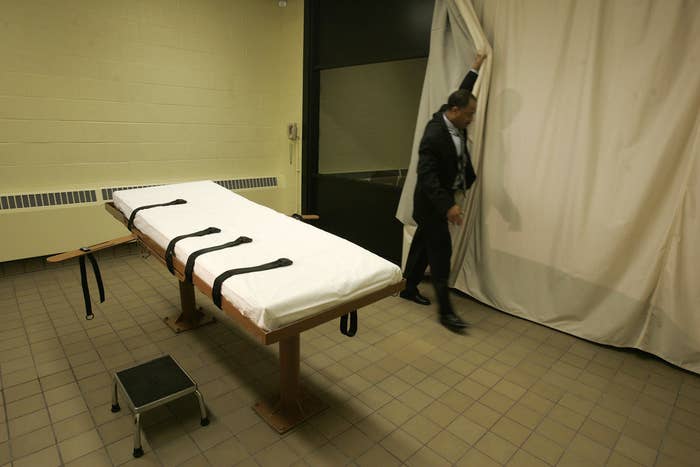
The prison Ohio carries out executions in registered for a DEA license to import the drug last year for a "law enforcement purpose," but until now it was unknown if the state actually intended to use the license.
Ohio, like many other death penalty states, shrouds its execution drug suppliers in secrecy. States argue the secrecy protects their suppliers from intimidation and embarrassment, while death row inmates and open government advocates argue it removes an important check on state power.
At this point, the identity of Ohio's intended supplier is unknown. Nebraska paid more than $50,000 for hundreds of executions' worth of sodium thiopental from an Indian dealer named Chris Harris. According to emails from Harris and the DEA, Harris sold to at least one other state as well.
When approached by BuzzFeed News about Harris in June, Ohio DRC spokesperson JoEllen Smith said the department's legal division would have to handle the matter. After spending weeks on the request, she only would say that Ohio had not communicated with Harris's company, Harris Pharma, but did not specifically answer the question of if the state had purchased from him directly or indirectly. Smith did not respond to follow up questions.
Ohio Gov. John Kasich and Attorney General Mike DeWine's offices would not answer questions either, deferring to the Department of Rehabilitation and Correction.
As many reputable drug options have dried up in recent years, states have become desperate, turning to questionable, and at times illegal, sources in attempting to carry out executions. Sodium thiopental's lone FDA-approved manufacturer stopped making the drug in 2011 in an attempt to keep the drug out of the hands of death penalty states.
Ohio has not carried out an execution since January 2014 — a 26-minute execution using different drugs in which the inmate gasped, snorted and appeared to struggle against his restraints. The state ditched that drug combination and is scheduled to begin a new round of executions in 2016.
Meanwhile, Nebraska has maintained that the state will still be able to import the drugs, in spite of the FDA's warnings, and in spite of the legislature repealing the death penalty this year.
In letters to Ohio, Nebraska, and Harris, the FDA directs officials to a 2012 court ruling that the FDA must refuse admission of sodium thiopental, as it is not approved by the drug agency. Before the ruling, the FDA had allowed several shipments in, arguing that FDA's oversight is limited to the medical field, not executions.
