
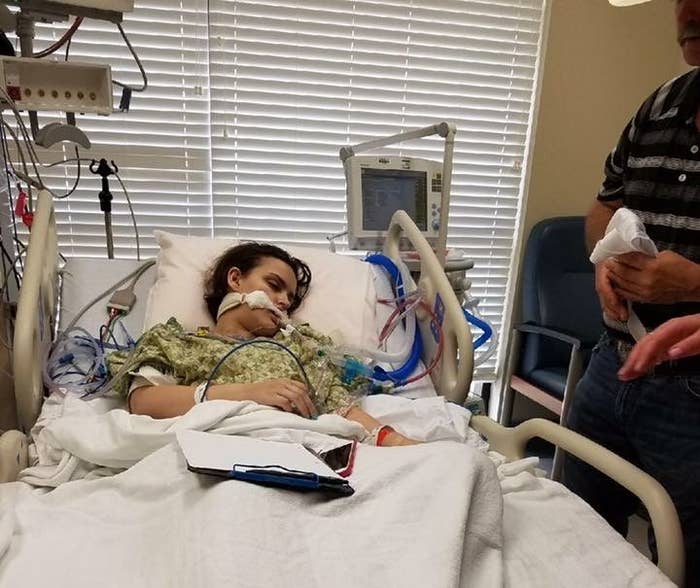
Paige Rogers had been connected to an IV at a hospital in Lubbock, Texas, for 25 days last year when her doctor came in with lab results showing she'd run out of options. The infection colonizing her lungs was no longer responding to antibiotics.
Rogers had been born with cystic fibrosis, a genetic disease that causes thick mucus to form in the lungs where bacteria can get trapped and multiply. The disease leads to persistent infections and harms the ability to breathe over time. The life expectancy of the nearly 30,000 people with the disease in the US is only 37 years old.
For more on this story, watch the new BuzzFeed News show Follow This on Netflix.
Since she was 2 years old, Rogers' lungs had been chronically infected by a type of bacteria called Pseudomonas. Since then, her life had been an endless cycle of getting sick, struggling to breathe, having countless doctors' appointments, and taking antibiotics to kill the bacteria. By the time she was 20, antibiotics were delivered straight into her blood through an IV every day, usually administered at home.
But last year, when she was 21, Rogers was checked in to the hospital for an extended stay because even with her medicine, she had started to feel weak and have trouble breathing. After surgery to try to remove some of the mucus in her lungs, she had to be intubated because she could no longer breathe on her own.
On day 25 of her hospital stay, her doctor confirmed her family's worst fears: The antibiotics she'd depended on for most of her life were no longer working. Rogers had become one of the millions of people in the US infected with so-called superbugs, bacteria that have evolved to thwart all of our existing antibiotics.
Her doctor said the best thing they could do was send her home.
"I wasn’t released because I was better — I was released because nothing was working," Rogers, now 23, told BuzzFeed News.
Desperate, her dad began searching online for any alternatives to antibiotics. He came across articles about a treatment that used live viruses to kill bacteria, only available in pockets of Eastern Europe. Her dad talked to some scientists in Russia, who then connected him to Benjamin Chan, a Yale scientist who was beginning to treat patients using phages in the US.
Chan is part of a growing community of patients, scientists, and doctors across the US fighting to make phage therapy more widely available. This effort is the subject of a new episode of the Netflix docuseries Follow This.
Rogers was initially worried about trying a treatment option that felt so uncertain, but was ultimately persuaded.
"I thought, there’s nothing else we can do now, and there’s probably no getting better, so I should at least try it," Rogers said. "I kinda felt numb. I didn’t feel happy and I didn’t feel scared to try them anymore either. But I didn't think it was going to help."
In June, her doctors and Chan began talking to the FDA about allowing Rogers to receive the experimental, unapproved treatment, given her lack of options. In November, her doctor sent a sample of mucus from her lungs to Chan, who began hunting for a virus that would kill the bacteria in her lungs.
These viruses, known as phages, aren't harmful to people; they only attack bacteria. There are trillions upon trillions of them on Earth — in soil, swamps, and anywhere else that bacteria naturally thrive.
Unlike antibiotics, which wipe out all bacteria in your system, phages only target specific strains of bacteria. So the key to using phages as treatment is finding the types that will optimally attack a patient's particular infection. In other words, phages are tailor-made to wipe out the bad bacteria making you sick, while leaving the good bugs alone.
When Chan received Rogers' bacterial sample, he immediately ran it against a collection of thousands of phages he had in his lab's library to see if he had anything that was a match. Chan has spent years collecting phages in sewage and lakes across the world. In 2016, he'd had success treating his first patient with phages he found in a lake in New England.
"Everywhere I go, I'm basically sampling," Chan said. "It's just a nearly limitless source, so we're just trying to collect as many as we can."

It took Chan about a month to find phages that were a strong match for Rogers' infection. He flew over to Lubbock to hand-deliver them to her, and her doctors began their treatment.
The phages were delivered to her body through a nebulizer, which turned them into a mist that could be breathed straight into her lungs. Rogers inhaled the vaporized viruses for 20 minutes to an hour each day.
The medicine worked slowly. But after 10 days or so, her doctors began to see a response. After another round of phages in February, Rogers' infection began to completely clear. And by March, she said, she felt better than she'd felt in years. Her lung function had drastically improved, and she felt strong enough to go back to work and live her life outside of a hospital.
She received another maintenance dose of phages this summer. Her lungs were not only still clear of the debilitating infection, but even better, the phages had weakened the bacteria in her system enough that they'd started responding to antibiotics again.
Few people in the US have been able to try phage therapy in the past few years, and it hasn't always worked. For example, the case of a 25-year-old cystic fibrosis patient named Mallory Smith made headlines in 2017 after a lung transplant left her besieged with an infection, but she died before scientists could find out if the phages worked.
Chan has worked with hospitals to treat five patients to date, and is helping some 50 others who have reached out looking for help. But as long as patients are treated on a case-by-case basis, he warns, the treatment will not reach everyone who needs it.
Chan is hoping to start a clinical trial testing phages on cystic fibrosis patients in the next year. Another group, at the newly founded Center for Innovative Phage Applications and Therapeutics (IPATH) at University of California, San Diego, is also planning to launch a clinical trial in the near future, in collaboration with private biotech companies. IPATH was launched after a UCSD professor of global health, Steffanie Strathdee, convinced the medical school to use phages to save her husband's life from a deadly superbug infection in 2016.
"I think in the next five years, this should be through clinical trials, and we should know for a fact whether it's worth pursuing, and whether we could actually help out a bunch more people with it," Chan said.
But challenges remain, including jumping through hoops with the FDA to approve living, custom-made medicines, as well as the economic hurdle of patenting a product that exists in nature.
Meanwhile, Rogers is back working part-time at a girls clothing store in her town. She's thinking of having kids, a possibility she'd never considered before she'd heard about phages. She breathes easier these days, she said, knowing her medical team can always find new phages if she gets sick.
"It’s crazy to think we were born with this disease that essentially takes a lot of people’s lives so young, and the lives that we do have are all about hospitals and doctors’ visits — they are lives but they’re not living," Rogers said. "I am excited about just being normal and getting to experience what average people take for granted." ●
